
Following recent accusations that the EMA puts companies' commercial interests ahead of public safety, the EMA has responded by emphasizing recent initiatives at the agency to improve data access.

Following recent accusations that the EMA puts companies' commercial interests ahead of public safety, the EMA has responded by emphasizing recent initiatives at the agency to improve data access.

FDA held a joint meeting of its Nonprescription Drugs Advisory Committee and Pediatric Advisory Committee to discuss whether new dosing information for oral over-the-counter drug products containing acetaminophen should be added to the label for children under age 2.

Almac opens new North American headquarters; Sanofi Pasteur appoints Damian Braga as senior vice-president of global commercial operations; and More.

Global spending on medicines will total almost $1.1 trillion by 2015, according to an IMS Institute for Healthcare Informatics study.

Teva Pharmaceutical Industries agreed to pay shareholders $460 million in cash to acquire a 57% stake in Taiyo Pharmaceutical Industry. Teva also will offer to buy all outstanding shares of Taiyo.

A bill was introduced in the US House of Representatives to launch a national strategy for the development of regenerative medicines.

Johnson & Johnson subsidiary Janssen-Cilag International reported that the company is working with regulatory authorities in five countries to address trace amounts of 2,4,6- tribromoanisole identified in five batches of the HIV/AIDS medicine Prezista.

AstraZeneca, GlaxoSmithKline, and the University of Manchester form new research collaboration for inflammation; BASi names Michael Zhou as senior director of R&D; and More.

EMA is working with its European and international regulatory partners to monitor and evaluate ?the possible risk of radioactive contamination of medicines manufactured in Japan following the radiation leak from the Fukushima Daiichi nuclear power plant.?

Amylin Pharmaceuticals has filed a lawsuit against Eli Lilly, alleging that Lilly is engaging in anticompetitive activity and breaching its agreements with Amylin to maximize commercialization of the drug, exenatide.

Regulatory approvals for new biopharmaceuticals in the United States have nearly doubled in the past decade compared with the 1990s, says a Tufts study.

Equipment and Processing Report
Recent recalls, including that of American Regent?s caffeine and sodium benzoate injection on May 5, 2011, highlight the importance of particulate inspection, and they might lead observers to ask whether current inspection methods are sufficiently effective.

Equipment and Processing Report
How do you assign a minimum sample weight for a US Pharmacopeia <41> balance application when the tested repeatability gives a standard deviation of zero?
Equipment and Processing Report
PharmTech's monthly newsletter, Equipment and Processing Report, reviews the Editor's Picks for the May 2011 edition from David Round Company and Globe Medical Tech.

Equipment and Processing Report
Filling active ingredients directly into capsules is probably the quickest option for entering clinical trials. This case study compares manual and automated methods of capsule filling.

The Office of the United States Trade Representative issued a report as part of its annual review of the global state of intellectual-property rights protection and enforcement.

In fiscal year 2010, the number of patent settlements in which the manufacturers of branded products paid makers of generic drugs to postpone the introduction of their products reached its highest level ever, according to the Federal Trade Commission.

FDA issued a new guidance for industry concerning the submission of summary bioequivalence data for abbreviated new drug applications.

FDA is asking for input on the development of a user-fee program for biosimilar and interchangeable biological product applications.

INC Research agrees to acquire Kendle; CMC Biologics hires Claes Glassell as CEO; and More.

Alkermes purchased Elan?s Elan Drug Technologies (EDT) unit in a cash and stock transaction worth approximately $960 million. The two companies will be merged into a new holding company in Ireland under the name Alkermes.

PhRMA Urges Congress to Reauthorize Legislation for Pediatric Drugs.

The IPEC is soliciting public comment about a draft plan for the independent certification of manufacturers and suppliers of pharmaceutical excipients.

Velesco partners with Vindonwestech; NeurogesX announces retirement of CEO Anthony DiTonno; and More.

A federal appeals court has lifted a ban on federal funding for embryonic stem cell research.

FDA is collecting public comments on a series of studies that the agency plans to conduct on online direct-to-consumer promotion of prescription drug products, according to an announcement in the Federal Register.

Israel-based Teva Industries seals deal to acquire Cephalon for $6.8 billion.

PTSM: Pharmaceutical Technology Sourcing and Management
Volume growth in the US prescription drug market was at historically low levels in 2010, and revenue growth was anemic.
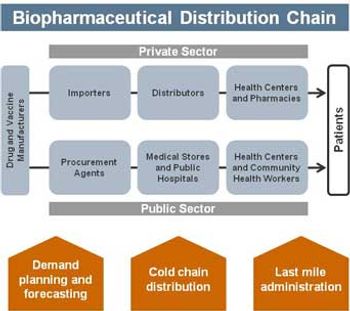
PTSM: Pharmaceutical Technology Sourcing and Management
This article, which focuses on distribution and administration, is Part III of a three-part series on biopharmaceutical issues in public health, government, and developing-world markets.

PTSM: Pharmaceutical Technology Sourcing and Management
A roundup of developments in corporate social responsibility and sustainability from the bio/pharmaceutical industry, its suppliers, and other public and private organizations.