
PTSM: Pharmaceutical Technology Sourcing and Management
GlaxoSmithKline and the Singapore Economic Development Board team in advancing sustainable pharmaceutical manufacturing.

PTSM: Pharmaceutical Technology Sourcing and Management
GlaxoSmithKline and the Singapore Economic Development Board team in advancing sustainable pharmaceutical manufacturing.

Pharmaceutical Technology Europe
As I strolled through Times Square, famous brand coffee cup in hand, towards the Jacob K. Javits Convention Center in New York, my thoughts were entirely focused on Interphex.

Eastern Europe's pharmaceutical leader, Hungary, is working to maintain its number-one status while also pursuing new avenues, especially in biopharmaceuticals.
Efforts are made to educate health workers in less developed countries.

Those who doubt there's faith in science, should check out our annual Bioprocessing Survey.

Can the semiconductor industry help Big Pharma develop therapies?

Monograph modernization and standards donation go hand in hand.

India has the potential to become the new star of the biotechnology industry.

Many factors affect research results.

Pharmaceutical Technology Europe
Despite initiatives to encourage multinational pharma companies to conduct R&D in the UK, the country may be losing its edge; is Pfizer's decision to exit a key site earlier this year the beginning of a mass exodus?

sanofi-aventis signs research pact with Stanford; Paul Maffuid joins AAIPharma Services; and More.

Warner Chilcott announced in a press release on Apr. 18, 2011, its intentions restructure, placing 500 Western European jobs on the line.

The IMS institute has released its report on the use of medicines in the United States during 2010.

Shanghai Pharmaceutical and Pfizer have signed a memorandum of understanding for the companies to jointly pursue potential business opportunities in China.

FDA has released a list of its strategic priorities for the next five years to address new global challenges.

The EMA has concluded a class review of bisphosphonates, adopted a number of positives opinions for new medicines and revealed more about the ongoing studies regarding GlaxoSmithKline's Pandemrix pandemic influenza vaccine and a possible link with narcolepsy.

GlaxoSmithKline (GSK) identified certain over-the-counter (OTC) brands in its consumer healthcare business that the company plans to divest.

The US patent laws are undergoing a major revision, the first large revision since the Patent Act of 1952.

Axcan acquires Mpex Pharmaceuticals; Bend Research receives patent for improving bioavailability of low-solubility drugs; and More.

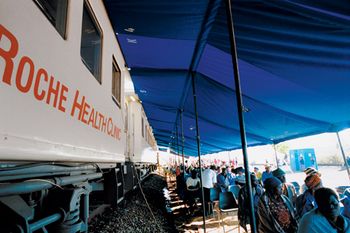
On Apr. 18, 2011, scientists from the national laboratories of five African nations gathered in Accra, Ghana, to take part in a week of technical training put on by the US Pharmacopeia (USP) that will teach them how to detect substandard and counterfeit medicines.

FDA Released Draft Guidance for Industry on Safety Labeling Changes.

On Tuesday, Apr. 19, 2011, officials from FDA, the US Department of Health and Human Services, and the Drug Enforcement Administration introduced President Obama?s plan for curbing prescription-drug diversion and abuse.

Equipment and Processing Report
The implementation of digital photomicrography has expanded the capabilities of microanalysis for quality control. But if used incorrectly, the technique can hurt more than help.

Equipment and Processing Report
We want to fill one of our oxygen-sensitive products inside an isolator system. Can we run an isolator with pure nitrogen and defined humidity?

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment and Processing Report, reviews the Editor's Picks for the April 2011 edition from ACS Valves and L.B. Bohle.

Equipment and Processing Report
To find out how well industry is applying QbD, and what benefits the approach can bring, Equipment and Processing Report talked to Moheb M. Nasr, director of FDA?s Office of New Drug Quality Assessment.

Alynlyam forms drug-delivery pact with the University of British Columbia; Linda S. Mayer Joins Schott North America; and More.

FDA recently published guidance for preventing the cross-contamination of finished pharmaceuticals and active pharmaceutical ingredients with nonpenicillin beta-lactam antibiotics.

Merck & Co. has formed a joint venture with the Mumbai-based specialty pharmaceutical company Sun Pharmaceutical Industries to develop, manufacture, and commercialize new combinations and formulations of branded generic drugs in emerging markets.

FDA Released Draft Guidance for Industry on Safety Labeling Changes.