
FDA cites Zhejiang Huahai Pharmaceutical in valsartan impurity investigation.

FDA cites Zhejiang Huahai Pharmaceutical in valsartan impurity investigation.

FDA has approved Truxima (rituximab-abbs), a biosimilar to Roche’s anti-cancer biologic, Rituxan (rituximab).
Pharmaceutical Technology's In the Lab eNewsletter
Integration of two separate chromatography data systems boosts workflow efficiency.

The proposal from the Centers for Medicare and Medicaid Services (CMS) proposes to give plans more flexibility to limit coverage of certain drugs.

A required time frame should not be the driving force behind root cause investigations, says Susan Schniepp, executive vice-president of Post-Approval Pharma and Distinguished Fellow, Regulatory Compliance Associates.
Microbial identity data can be critical for determining contamination sources.

Bio/pharma companies cannot spell success without solving all elements of quality programs.

Success depends on supplier communication and transparency, but it’s up to buyers to demand the right information and to look at the vendor’s overall business goals.

Simplified role-based training can lead to better quality metrics and compliance.

Once described as “throwing processes over the wall,” tech transfer is evolving into close collaboration and communication, as potential problems are considered sooner, and new technology is applied. Joseph Szczesiul, director of technical services for UPM Pharmaceuticals, shares best practices.

Drawing on practical experience, the authors examine key questions and answers about various aspects relating to the enhanced approach for analytical procedure lifecycle management.

EMA’s relocation to Amsterdam and resulting staff losses could severely weaken the agency’s role as a leading medicines regulator.
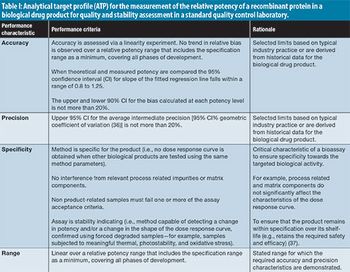
Three hypothetical analytical target profiles (ATPs) are provided, reflecting the current thinking of the the European Federation of Pharmaceutical Industries and Associations Analytical Lifecycle Management Team.
GMP non-compliance can spill over and impact patient access to life-saving medications.

Owner of Immuno Biotech, David Noakes, has been sentenced to 15 months in prison over charges of manufacturing, selling, and supplying an unlicensed medicine, as well as money laundering.

Five additional European Union member states have been confirmed by the US Food and Drug Administration (FDA) as capable of performing good manufacturing practice inspections at a level equivalent to that of the United Sates

Pharmaceutical Technology's In the Lab eNewsletter
A new, high-throughput microplate reader cuts down on screening time and works faster than standard ultra-high-performance liquid chromatography processes.

Pharmaceutical Technology's In the Lab eNewsletter
The partnership, co-funded by Enterprise Ireland, will develop technologies for monitoring the quality of biopharma processes.

Despite ongoing efforts to address the problem, FDA now sees a rise in active shortages and in the duration of supply problems.

The agency has approved two new chemical entities, Daurismo (glasdegib) from Pfizer, and Vitrakvi (larotrectinib) from Loxo Oncology, for treating cancers.

FDA has issued a warning letter to Mylan citing GMP violations of finished pharmaceutical products manufactured at the company’s Morgantown, WV, facility.

The positive opinions included the first oral-only tablet for the treatment of human African trypanosomiasis.

After a review of serious side effects, the agency decided to suspend marketing authorizations for quinolone and fluoroquinolone antibiotics and restrict use of remaining fluoroquinolone antibiotics.

Moving forward with gene therapy development requires a “quantum leap” in manufacturing capabilities.

The agency provided an update on its relocation plans and assured that core activities are continuing uninterrupted.