
FDA officials said on Feb. 5, 2016 that Celltrion’s biosimilar to infliximab was “highly similar” to Johnson & Johnson’s (J&J) Remicade, according to a report by Reuters.

FDA officials said on Feb. 5, 2016 that Celltrion’s biosimilar to infliximab was “highly similar” to Johnson & Johnson’s (J&J) Remicade, according to a report by Reuters.

AMETEK announced the acquisition of Brookfield Engineering Laboratories, a Massachusetts based manufacturer of viscometers and rheometers.

Although switching has occurred in European markets for some biosimilars, most biosimilar manufacturers will focus on securing new users, according to Merck.

Sartorius releases financial results for 2015, announcing a 16% increase in group sales revenue.

ViaCyte and Janssen Biotech have entered Phase I/Phase II clinical trials for VC-01, a candidate treatment for the treatment of type 1 diabetes.

In a hearing held on Feb. 4, 2016, executives from Valeant and Turing had a hard time explaining their rationales for exorbitant price hikes of older drugs.

LabConnect built a biorepository facility in Tennessee.

AstraZeneca received conditional marketing authorization for Tagrisso, a tablet for the treatment of adults with locally advanced or metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer.
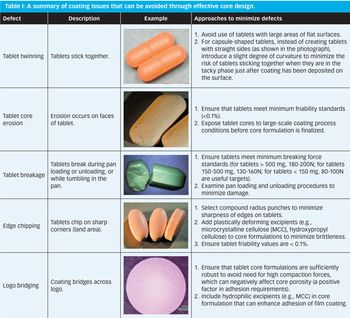
Common defects in tablet film coating can be minimized by effective design of the tablet core and the coating process.

Catalent Pharma Solutions’ technical project manager, Elanor Pinto-Cocozza, PhD, will present at InformEx 2016.

Systag, a Swiss fine chemicals and process automation company, appeared at InformEx 2016

InformEx, a conference for the fine and specialty chemicals industry, kicked off on Feb. 2, 2016 in New Orleans.

Several biopharmaceutical companies announce plans to launch R&D into Zika vaccine candidates.

The International Society for Pharmaceutical Engineering (ISPE) Facility of the Year Awards (FOYA) program announced its 2016 Category Award winners for operational excellence, sustainability, process innovation, project execution, equipment innovation, and facility integration.

Suppliers must develop new technologies to drive the bio/pharma innovation engine.

Catalent plans a $4.6 million investment to expand secondary packaging and storage in Asia.

The chemical distribution industry has formed an international chemical trade association to address global issues.

Can the feds negotiate Medicare Part D prices?

The Biosimilars Forum launched Partnership for Biosimilars Education and Access, an education initiative raising awareness of biosimilars in the US.

Pfizer and Bristol-Myers Squibb enter into agreement with Portola to develop and commercialize andexanet alfa in Japan.

CDER’s Office of Pharmaceutical Quality plans on promoting modernization as a way to ensure drug quality.

Aging populations and increased access to healthcare translates into opportunities for biopharmaceutical companies.

FDA and industry seek a more consistent, flexible CMC review process for breakthrough therapies.

PTSM: Pharmaceutical Technology Sourcing and Management
Hermes Pharma has commercially implemented hot-melt coating (HMC) technology at its production facility.

PTSM: Pharmaceutical Technology Sourcing and Management
Novasep is building a new synthesis laboratory and adding capacity for kilogram-scale batches of synthetic molecules that are needed for biological testing and preclinical trials, at its Pennsylvania, US facility.

Novartis announced the companies plan to boost growth at Alcon, the company’s vision care division.

The agency is seeking industry input on product-specific recommendations to facilitate generic drug development.

Amgen announces FDA will review the company’s BLA for ABP 501.

NICE announces plans to back biosimilar alternatives to Merck’s Remicade.

On Jan. 27, 2016 FDA announced it plans to review Merck’s investigational antitoxin prevention bezlotoxumab.