
Members of the House Energy and Commerce Committee are expanding their investigation into the 2008 entry of contaminated heparin from China into the United States market.

Members of the House Energy and Commerce Committee are expanding their investigation into the 2008 entry of contaminated heparin from China into the United States market.

Sanofi Forms Research Pact with Weill Cornell Medical Center; Steven A. Nichtberger Resigns as President and CEO of Tengion; and More.

Getting an answer is easy-asking the right question is apparently more difficult.

A new report places pharmaceutical and healthcare companies ahead in corporate and social governance.

GSK Works in Developing Nations to Improve Everything from Research to Roads.

Member states in the EU are working to implement the newly passed Falsified Medicines Directive.

US support of regenerative medicine is essential for maintaining a lead on healthcare innovation.

Which route will we take to arrive at a national stem-cell policy?

Pharmaceutical Technology Europe
Switzerland is an important power in Europe's pharmaceutical industry.

Pharmaceutical Technology Europe
One of the perks of my job is that I can travel to previously unvisited cities. This month, I was lucky enough to be in Switzerland.

FDA Publishes Final Guidance on Dissolution Testing.

Roche Establishes R&D Institute in France; Sanofi Appoints Peter Guenter as Senior Vice-President, Europe; and More.

President Obama unveiled an Advanced Manufacturing Partnership designed to reinvigorate the country's manufacturing sector.

Industry analysts have highlighted the pharma industry's high rate of Phase III drug terminations and significantly reduced R&D spending.

The IPEC Federation has issued a statement on the use of phthalates in pharmaceutical products in response to reports of adulteration of certain nutritional supplements, vitamins, foods, and beverages imported from Taiwan.

The European Medicines Agency (EMA) has held a second forum regarding the implementation of new pharmacovigilance legislation, which gave stakeholders the opportunity to discuss their expectations on various aspects of the new legislation's execution.

The Association of the British Pharmaceutical Industry (ABPI) has published a guidance that suggests best practices for managing adverse events and other pharmacovigilance data from the internet and social media tools.

FDA Issues Warning Letter to Dr. Reddy's Following Inspection of the Company's Mexico-Based API Manufacturing Plant.

The traditional method of conveying information in the brief summary of a printed prescription-drug advertisement is neither the most comprehensible nor the most preferred by consumers, according to an FDA study.

FDA released a new strategy on that is aimed at meeting the challenges posed by rapidly rising imports of FDA-regulated products and the growing complexity of the pharmaceutical supply chain.

Array BioPharma Restructures to Focus on Development of Clinical Programs; AstraZenica Plans Russian Expansion; and More.

A subsidiary of Johnson & Johnson initiated a voluntary recall of its antipsychotic medication, Risperdal, due to an unusual odor.

Novartis has commenced construction of a new manufacturing plant in Russia that represents the company's most significant investment in the country to date.

Biogen Idec Receives Approval for the Avonex Pen; Xceleron Makes Several Senior Appointments; and More.

GlaxoSmithKline has entered into an agreement to purchase Shenzhen Neptunus? stake in a previously formed joint venture between the companies involved in the development and manufacture of influenza vaccines in China, Hong Kong, and Macau.

FDA Issues Consent Decree of Condemnation, Forfeiture, and Permanent injunction Against H&P Industries, the Triad Group, and Three Individuals.

Merck and Hanwha Chemical have formed an exclusive global agreement to develop and a commercialize a biosimilar of Enbrel, a drug to treat moderate to severe plaque psoriasis, psoriatic arthritis, and moderate to severe rheumatoid arthritis.

An FDA guidance has provided information for products that involve the application of nanotechnology, and rationales for those points.

After looking back at the first year of its Bad Ad outreach program, FDA judged that the initiative has successfully raised awareness about misleading promotion, according to an FDA press release.
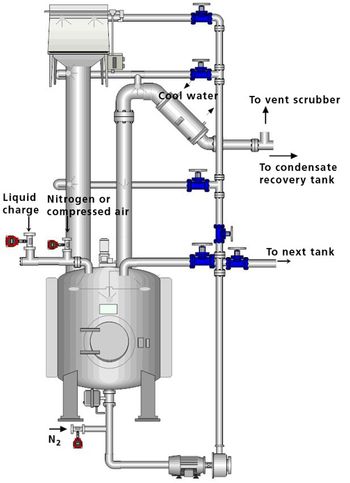
Equipment and Processing Report
The author describes the benefits and challenges inherent to cleaning in place (CIP). The article also describes the development and validation of a CIP cycle.