
Myth versus Reality: What does Q10 implementation really mean for my company?

Surrounded by competition, Vietnam's 2020 vision focuses on building a biotech sector worthy of its Asian neighbors-as well as the growing global biopharmaceutical market

Legislative decisions to increase Medicare's formulary may lead to a fight over drug approvals.

USP's guideline for pending monographs can speed up publication of monograhs and time to market.

Despite its flaws, a recent release fills a need for books about pharmaceutical project management.
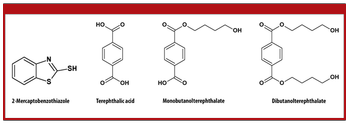
Extractables and leachables are a growing concern for pharmaceutical manufacturers and regulatory bodies.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency recommended strengthening the warnings for oral moxifloxacin medicines and concluded that these drugs should only be prescribed to treat acute bacterial sinusitis, acute exacerbation of chronic bronchitis, and community-acquired pneumonia when other antibiotics cannot be used or have failed.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Cobra Biomanufacturing to extend collaboration and form joint venture with ViroMed, Epix Pharmaceuticals CEO resigns, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pharmaceutical companies developing new drug candidates for Hepatitis C virus infection now can test their compounds with a novel culture system that mimics the biology of HCV infection in humans.

ePT--the Electronic Newsletter of Pharmaceutical Technology
As part of a new strategic direction, GlaxoSmithKline CEO Andrew Witty outlined three priorities that are designed to improve the company's long-term financial performance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Senator Sherrod Brown sent a letter to Richard T. Clark, president and chief executive officer of Merck (Whitehouse Station, NJ), to ask for information about the company's reliance on global outsourcing for the manufacture of pharmaceutical ingredients and finished products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration seeks applicants for its new Commissioner’s Fellowship Program for scientists, engineers, and health professionals. The two-year program will help prepare the agency to replace a large number of FDA staff who are preparing to retire as well as meet future goals.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Teva Pharmaceutical Industries and Barr Pharmaceuticals (Montvale, NJ) signed a definitive agreement under which Teva will acquire Barr, the fourth-largest generic-drug company in the world. Teva will buy each share of Barr common stock for $39.90 in cash and 0.6272 Teva shares. Based on the closing price of Teva?s shares on July 16, 2008, the price per share of Barr common stock is $66.50, and the total value of the acquisition is $7.46 billion. Teva will assume Barr?s outstanding net debt of approximately $1.5 billion.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Roche plans to acquire the biopharmaceutical company Genentech (South San Francisco, CA) for $43.7 billion. Roche currently holds a 55.9% stake in Genentech and it plans to acquire the remaining publicly held minority interest that it does not own for $89 per share.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Niro changes its name to GEA Process Engineering; Frances K. Heller joins Exelixis as executive vice-president of business development, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued a final rule on July 18 that exempts early Phase I investigational drugs from certain good manufacturing practice regulations.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer (New York, NY) announced plans to restructure staff at its Kalamazoo, Michigan, facility, leading to an estimated job cut of 275 by the end of the year.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Bristol-Myers Squibb (BMS) agreed to reduce the output of ozone-depleting refrigerants at several industrial facilities around the country to resolve violations of the Clean Air Act. The company's modifications will cost approximately $3.65 million.

ePT--the Electronic Newsletter of Pharmaceutical Technology
3M Drug Delivery Systems has successfully designed a proof of concept device using a solid microstructured transdermal system for the systemic delivery of high-potency pharmaceuticals. The technology was showcased at a poster session at the annual meeting of the Controlled Release Society held this week in New York City.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Roche to end HIV/AIDS research program, WuXi PharmaTech makes appointments, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration published a Draft Guidance for Industry on Providing Regulatory Submissions in Electronic Format in the Federal Register on July 11.
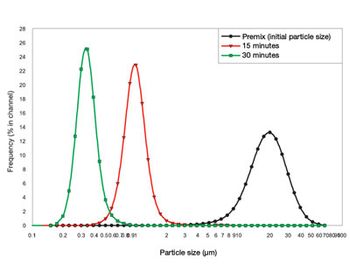
Equipment and Processing Report
Creating nanoparticles can be challenging and requires appropriate equipment and techniques. Media milling or grinding is the best-established manufacturing method for nanoparticle production.

Equipment and Processing Report
A pharmaceutical company's information-technology infrastructure could facilitate the appraisal of the performance of contract manufacturing organizations.

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment & Processing Report, reviews the Editor's Picks for the July 2008 edition from Parker Fluid Control Division and Watson-Marlow Bredel.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer has agreed to pay $975,000 in civil penalties to resolve alleged violations of the Clean Air Act at its former manufacturing plant in Groton, Connecticut, according to a release by the US Department of Justice.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Catalent Pharma Solutions to collaborate with One World Design and Manufacturing Group, Bioheart appoints Howard J. Leonhardt as CEO, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration is seeking volunteers from the pharmaceutical industry to participate in a pilot program involving the submission of quality information for biotechnology products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Pharmacopeia Commission has published the General Information chapter "Potentially Genotoxic Impurities and European Pharmacopoeia Monographs on Substances for Human Use" in the July 2008 edition of PharmEuropa.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Pharmacopoeia Commission adopted revised monographs for heparin calcium and heparin sodium to strengthen the level of testing required for quality control.

PTSM: Pharmaceutical Technology Sourcing and Management
Change control in life-sciences organizations is a critical business issue in terms of risk, safety, and performance. The author examines common shortcomings in change control when implementing non-enterprise solutions and the functionality derived from enterprise-level change control.