
Data capture needs to be fast and reliable...so which automatic identification technology is best?

Data capture needs to be fast and reliable...so which automatic identification technology is best?

Attendees at a recent workshop endorsed new methods to detect metals in drugs, dietary supplements, and food ingredients.
With government support, China's pharmaceutical equipment sector is trekking ahead despite challenges regarding the country's overall perceived product quality.

The Indian government may soon monopolize its pharmaceutical industry to cut costs and improve healthcare, but the move is sounding off alarm bells with the companies whose drug products are under review.

The US Food and Drug Administration seeks to understand nanotechnology better and exercise appropriate oversight over products that incorporate it.

Pharma companies could benefit from the lessons learned in this fall's financial crisis.

Production problems come in all shapes, sizes, and ... species.

IPEC Chairman Dave Schoneker discusses current efforts toward facilitating regulatory reviews of new excipients.


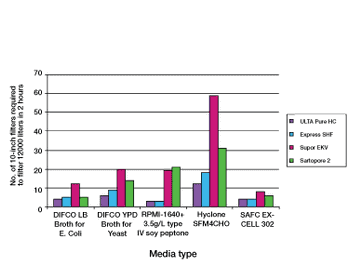
Proper selection of normal flow filters leads to increased process efficiency from early phase product development through to full-scale biopharmaceutical production.

A comparison of conventional cleanrooms, restricted access barrier systems, and isolators, shows the benefits of using isolators in high-potency drug manufacturing.

Scott Sutton discusses the current state of USP ‹1117› and USP's plans for future revisions.

Pharmaceutical Technology Europe
The first 2 weeks of October witnessed the closure of CPhI (30 September–2 October) and the whole of Biotechnica (7–9 October). Pharmaceutical Technology Europe had a presence at both events, which took place in Frankfurt and Hanover (both Germany), respectively. With record-breaking numbers of attendees, roundtables and conferences of the highest quality, and exhibitors from bio and pharma companies from all over the world, these two events are a must in the calendars of many in the biotech and pharmaceutical industries.

Pharmaceutical Technology Europe
Together, Europe and the US account for more than 70% of the global pharmaceutical market, and the growth of these markets is heavily dependent on distribution systems.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Government Accountability Office (GAO) released a report last week that examines and provides recommendations for the US Food and Drug Administration's foreign inspection process, including the agency's data management, inspection frequency, and oversight of problems identified during inspections.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration has awarded the National Institute for Pharmaceutical Technology and Education (NIPTE), a not-for-profit organization comprising 11 universities, a $1.19 million contract to develop quality by design (QbD) guidance elements for design space and scale-up of unit operations.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Merck plans to cut approximately 7200 positions as part of its 2008 restructuring plan, according to an Oct. 22 release focused on the company's third-quarter financial results.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer and Lilly recently resolved issues related to their marketing practices for certain products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Maxygen looks to costs, jobs; Receptor BioLogix appoints Dale R. Pfost CEO; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer and UCB formed a technology company named Cyclofluidic with the aim of accelerating the drug-discovery process.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration released a draft guidance that reviews the agency's plan to offer priority-review vouchers to companies developing new treatments for neglected tropical diseases.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Department of Health and Human Services (HHS) will send the first Food and Drug Administration staff to China, India, Europe, and Latin America before the end of 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Rep. John D. Dingell (D-MI), chairman of the US House of Representatives Committee on Energy and Commerce, and Rep. Bart Stupak (D-MI), chairman of the Oversight and Investigations Subcommittee, sent letters to the US Food and Drug Administration, Shaw Science Partners, and EthicAd to request information about a new FDA website.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, MedImmune opens Cambridge, UK, facility and makes reverse engineering pact with Omninvest; BD Medicine appoints Carol Adiletto VP of clinical and regulatory affairs; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
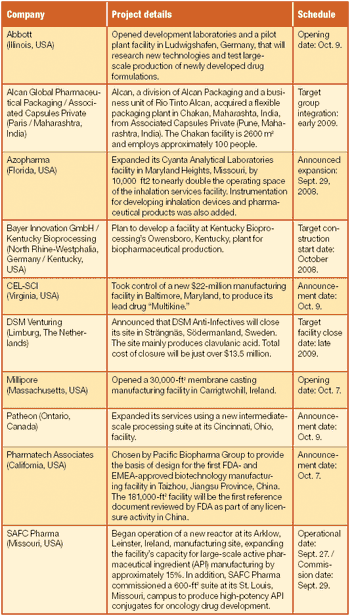
Also, Millipore opens new membrane-casting manufacturing facility in Ireland; Surface Logix appoints Keith Dionne president, CEO, and a member of the board; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
According to a July 11, 2008 memorandum posted by the Center for Biologics Evaluation and Research, starting Oct. 15, Health Level 7 structured product labeling in XML (extensible markup language) will be the only acceptable presentation in electronic format for the submission of content of labeling that CBER can process, review, and archive.

ePT--the Electronic Newsletter of Pharmaceutical Technology
FDA has completed its labeling updates to fluoroquinolone antimicrobial drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
During the past several months, Pharmaceutical Technology has been covering the US Food and Drug Administration's rulemaking on over-the-counter (OTC) cough and cold medications for children.