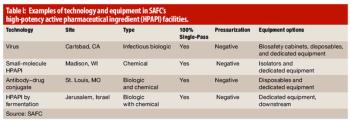
High-potency manufacturing of active pharmaceutical ingredients is a growing and specialized capability.

High-potency manufacturing of active pharmaceutical ingredients is a growing and specialized capability.

Traditional Chinese Medicine is widely used, but questions persist regarding its regulatory status.

A look at the true cost-drivers of cell-culture production.

Developers of low-dose drugs in solid oral dosage forms will find theoretical considerations and practical advice in a new book.
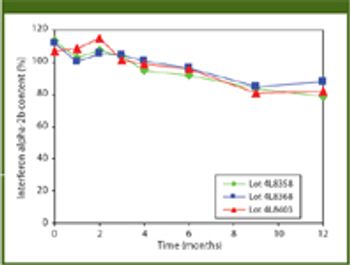
The authors describe a proprietary process for producing a stable, topical interferon alpha-2b formulation that can deliver large drug molecules into the skin or mucosa.
Pharmaceutical Technology Europe
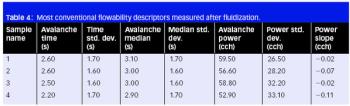
Cefaclor is a β-lactam cephalosporin antibiotic that has a wide particle size distribution. Because of the nonporous nature of the material, the specific surface area value accounts for a significant amount of fine particles possibly present in the samples under analysis.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Adimab forms deals with Merck and Roche; Manhattan Pharmaceuticals' CEO and president steps down; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA debars clinical investigators; Jubilant Organosys forms deal with Endo Pharmaceuticals; AMRI makes changes to its India management team; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Subcommittee on Health of the US House of Representatives Energy and Commerce Committee held hearings last week to discuss the findings of a report by the Federal Trade Commission (FTC) that examined the competitive effects for follow-on-biologics (FOBs).

Equipment and Processing Report
The emergence of influenza A (H1N1) and the efforts to provide vaccines to the vulnerable are timely examples of biopharmaceuticals' continuing importance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, TorreyPines Therapeutics to liquidate assets and dissolve company; EU's competition services to examine Pfizer/Wyeth merger; Akorn appoints Raj Rai interim CEO; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Commission's (EC) Directorate-General for Enterprise and Industry (EC-DG Enterprise) announced last week that it will not continue preparing a commission directive on good manufacturing practices (GMPs) for certain excipients.

PTSM: Pharmaceutical Technology Sourcing and Management
Consumer-care products, electronics, and select industrial firms comprise AMR Research's Supply Chain Top 25 for 2009, offering an opportunity for the pharmaceutical industry to examine best practices in supply-chain management.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, WACKER expands Iowa facility; EMEA releases a Q&A document for PIPs; Metrics consolidates quality operations; more...

The authors describe the importance of a rapid and an abbreviated screening strategy in initial solvent screening. This article contains bonus online-exclusive material.
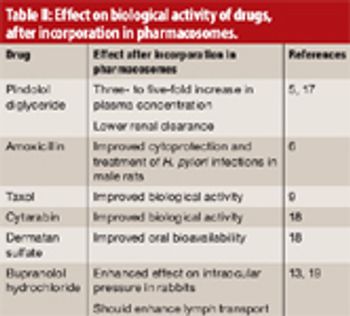
Pharmacosomes can pass through biomembranes efficiently and possess several advantages over traditional vesicular drug-delivery systems.

Misleading the public about their investments-be it money or medicine-is unacceptable.

CROs and CMOs expand to gain a piece of the market for clinical trial materials.

Follow-on biologics could unleash the potential of several industries and may even spark economic recovery.

It can take a lot of work to make sure nothing happens.

Short-term problems in software or hardware lead to long-term manufacturing troubles.
Pharmaceutical Technology Europe
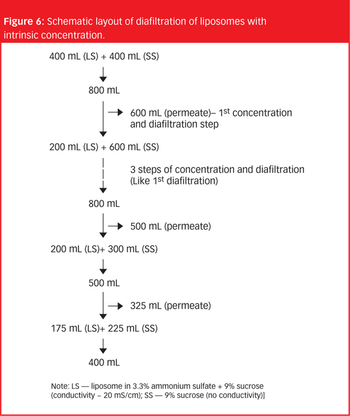
The importance of liposomes as an effective drug delivery system is well accepted in the pharmaceutical industry, but their handling remains a challenge.

Pharmaceutical Technology Europe
In 2005, a small delegation (myself included) of the European Fine Chemicals Group (EFCG) met with the deputy head of the cabinet of Commissioner Kyprianou (the then Commissioner responsible for health and consumer protection). Our mission was simple - we were there to raise a red flag.

Pharmaceutical Technology Europe
Today, approximately 1.5 million counterfeit medicine packs enter the legal supply chain each year - in other words, one pack in every 20000 is counterfeit.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Johnson & Johnson acquires Cougar Biotechnology; NIH launches program for rare and neglected diseases; PPD restructures leadership positions; more...