
ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Pall plans expansion in South America, Anthony Clarke joins Alexza Pharmaceuticals, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Pall plans expansion in South America, Anthony Clarke joins Alexza Pharmaceuticals, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration advised patients, caregivers, and healthcare professionals to switch to hydrofluoroalkane-propelled albuterol inhalers now because chlorofluorocarbon-propelled inhalers will not be available in the US after Dec. 31, 2008.

Antibody drug conjugates offer a niche opportunity in drug development and contract manufacturing.
Creating a kinder, gentler manufacturing process that doesn't kill the product is the goal of process developers doing large-scale cell culture for cell therapy.

Nanoparticle-based systems present many advantages for the delivery of current and emerging biological drugs.

Letting the public inside the drug development process may increase their faith in what we do.

The good, the bad, and the ugly about direct-to-consumer advertising.

Scientists are uncovering signaling systems that operate via cannabinoid messenger molecules.

An ambitious survey of characterization techniques presents current information.
Pharmaceutical Technology Europe
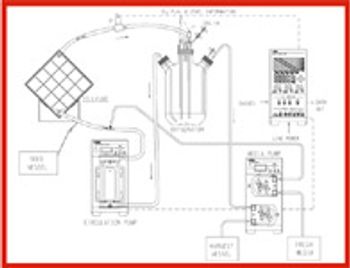
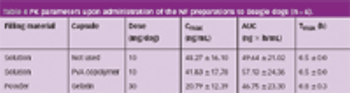
When drugs are encapsulated, electrification (the electrostatic charge of the capsule) may sometimes cause problems, such as capsule adhesion during transportation or dispersion of the capsule content in the filling process.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, GVK BIO and Wyeth Pharmaceuticals form research agreement, Eli Lilly announces changes to management, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Federation of Pharmaceutical Industries and Associations (EFPIA), the trade association representing European pharmaceutical manufacturers, issued recommendations to the public consultation launched in March by the European Commission's proposed drug anticounterfeiting measures. EFPIA's proposal includes a ban on drug repackaging.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Pfizer to close Indiana "Exubera" facility, executive appointments at Patheon, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Quintiles Transnational to acquire Eidetics, ChemAxon appoints Alex Drijver CEO, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA removes OAI status for Watson's Florida facility, executive management changes as GSK, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Polyplus-transfection, a company that researches, develops, and commercializes drug-delivery solutions for biomolecules, created a new technology designed to enhance in vivo delivery of small interfering RNAs (siRNAs) when they are associated with a cationic polymer.
Scientists are giving up on a preventive vaccine for AIDS, but there are lessons to be learned.

An authoritative book helps drug developers face one of their toughest problems.

The less complex nature of excipient manufacturers, as compared with API manufactures, carries many benefits.
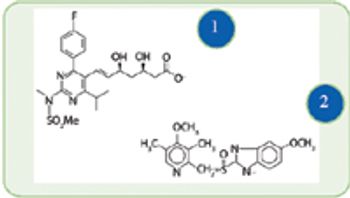
Chemocatalytic and biocatalytic routes show promise for more efficient syntheses of select active ingredients.

Pharmaceutical Technology Europe
Insulin is one of the world's oldest and most well-known biological drugs, and the need for it is not going to go away as the number of patients diagnosed with diabetes continues to increase. A wealth of clinical evidence shows that good, long-term glucose control in diabetes is key to avoiding complications such as kidney disease, blindness and heart problems.

The current trend within the pharmaceutical industry toward more efficient development, manufacturing, and specification is fueling demand for analytical tools that provide highly relevant information. Effective powder characterization has a valuable role to play.
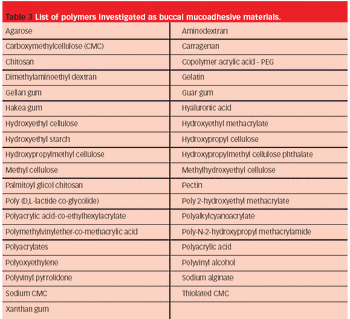
Pharmaceutical Technology Europe
The adequate absorption and transport of drugs in the body is part of optimal therapy. Drug administration perorally is easy, common and traditional, but occasionally alternative routes are required.

Pharmaceutical Technology Europe
The assembly of the World Health Organisation (WHO) later this month is expected to feature counterfeit medicines as one of its important discussion points, and there is some hope that the ministers will agree on measures that will strengthen anticounterfeiting legislation and enforcement worldwide.
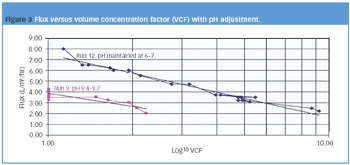
Pharmaceutical Technology Europe
The pharmaceutical industry must address the release of nonbiodegradable APIs into the environment.