Quality Systems
Latest News
Latest Videos

More News

This article outlines common CMC problems that are obstacles that steal momentum and create costly detours in the development or orphan drugs.

The European Union aims to become the most attractive place for life science by 2030.

The increasing diversity and complexity of injectable drug products is driving innovation.
Pharmaceutical Technology® spoke with Juliana Maynard, PhD, Head of Translational Imaging at Medicines Discovery Catapult, to find out what makes radiopharmaceuticals unique and how MDC’s collaboration with Crown Bioscience can help developers of these treatments for cancer.

Siegfried Schmitt, PhD, vice-president, Technical at Parexel, answers questions on the use and benefits of real-world evidence for small-molecule and large-molecule drug development.

This review of the nitrosamines contamination problem in pharmaceuticals takes a look at how the crisis started and developed.

A draft reflection paper on patient experience data is up for public consultation until Jan. 31, 2026.

FDA's Center for Biologics Evaluation and Research has released updated draft recommendations for sponsors of cell therapies, gene therapies, and tissue products.

In this episode of the Ask the Expert video series, Susan J. Schniepp, Regulatory Compliance Associates, and Siegfried Schmitt, PhD, Parexel, answer questions on the use of real-world evidence for both small-molecule and large-molecule drug development. In addition, they tackle a question on supply chain security problems that arise during transportation of pharmaceutical goods.

The FDA draft guidance "Considerations for Complying with 21 CFR 211.110" raises points to consider regarding drug products made using advanced manufacturing, batch uniformity, drug product integrity, and how manufacturers can incorporate process models into control strategies.

The dispute shines a light on the vulnerability of long-established drugs to renewed safety scrutiny: Even when causal evidence is lacking, observational findings can influence policy and public perception.

Advancements for novel cancer therapies are booming. This video takes a look at some of the latest efforts in the cancer research and therapy arena.

EMA states the new variations guidelines will streamline lifecycle management and make processing variations quicker.
CDC panel shifts vaccine policy by discouraging combined MMRV shot for young children due to increased febrile seizure risk.

Pharmaceutical Technology® spoke with Todd Sprouse, associate director, Analytical Services, and Erik Feldmann, PhD, principal technical advisor, Client Solutions & Proposals, both with Cambrex, to find out more about the challenging situation.
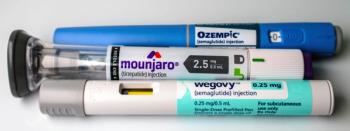
Misleading promotions of GLP-1 and compounded semaglutide products are drawing renewed regulatory scrutiny over risk disclosure and safety messaging.

Bram Baert, global head of Regulatory Affairs at Lonza CHI, gives his perspective on the impact of the EC’s decision on the use of TiO2 in drug products.

Susan J. Schniepp, Distinguished Fellow, Regulatory Compliance Associates, joins PDA CEO Glenn Wright and Melissa Seymour, Executive Vice President, Global Quality, Eli Lilly and Company, to talk about the regulatory and quality topics discussed at the 2025 PDA Regulatory Conference.

Charles Gibbons of Lachman Consultants and Michael Grischeau of AbbVie stressed AI governance, data integrity, and human oversight as essential to applying digital tools across labs and supply chains.

The performance of ARS Pharmaceuticals’ Neffy marginally outpaced historical reports of epinephrine injection effectiveness.

The agency says it will now publish complete response letters promptly, offering developers early insight into regulatory deficiencies to improve development planning.

The nonprofit is calling upon its community of experts in both artificial intelligence and machine learning to continue building support for responsible adoption of AI in the bio/pharmaceutical industry.

Digital solutions help pharmaceutical manufacturers navigate the complex supply chain.

This article previews some formulation topics discussed in the Pharma Fundamentals series, which aims to enhance learning for professionals in pharmaceutical drug development and manufacturing.

Michelle Heiter, laboratory and quality control specialist, and Susan J. Schniepp, distinguished fellow, Regulatory Compliance Associates, a Nelson Labs Company, discuss how to effectively use contamination control strategies in your operations.