
ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention announced a revised glycerin monograph in the United States Pharmacopeia.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention announced a revised glycerin monograph in the United States Pharmacopeia.

ePT--the Electronic Newsletter of Pharmaceutical Technology
As part of ongoing efforts to improve the quality of imported drug products, the US Food and Drug Administration is setting up shop in China.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Pipex Pharmaceuticals implements cost-cutting measures, Pfizer's Senior Vice-President and General Counsel Allen Waxman leaves the company, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Senate passed an amendment to the fiscal year 2009 budget bill that will increase the amount allotted to FDA by $71 million, bringing the agency's total FY 2009 budget increase to $375 million.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Hoping to provide a better understanding of its role in the heparin contamination investigations, the US Food and Drug Administration has posted a series of information sheets on its website.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention (USP) signed a Memorandum of Understanding (MOU) with the Chinese Pharmacopoeia Commission (ChP) designed to strengthen the quality of medicine and foods in the United States and China.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US House of Representatives Committee on Homeland Security approved legislation last week that mandates inherently safer technologies (IST) as part of chemical-site security standards, a move that was opposed by the Synthetic Organic Chemical Manufacturers Association.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Concerns over contaminated heparin product worsened as US officials announced dialysis patients in Germany have fallen ill after using a different brand of the blood thinner than was already being recalled.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, PDL BioPharma will no longer pursue sale of the company, executives resign from Topigen Pharmaceuticals, more...

Much to the surprise of most close observers of the Food and Drug Administration, Janet Woodcock agreed last month to resume control of the Center for Drug Evaluation and Research.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Millipore plans to open Singapore facility, Michael J. Simms joins Alexza Pharmaceuticals, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
With another potential "made-in-China" crisis looming over the recall of heparin, critics and the media seem to be waiting in line to take another jab at the US Food and Drug Administration.

PTSM: Pharmaceutical Technology Sourcing and Management
Draft federal legislation that would require high-risk chemical facilities to use inherently safer technology for reducing their risk may present potential problems for custom and batch manufacturers supplying the pharmaceutical industry.

There are significant differences between the United States and European Union requirements for the annual review of records related to the manufacturing and control for pharmaceutical products and active pharmaceutical ingredients.

Quality-by-design submissions may reduce supplements and improve change management.

A. Nair discusses patent disputes in India.

If not properly monitored, filters and plastic bags can keep back more than they should.

The United States Pharmacopeia emphasizes mechanical calibration and a performance test to esnure integrity of the dissolution procedure.
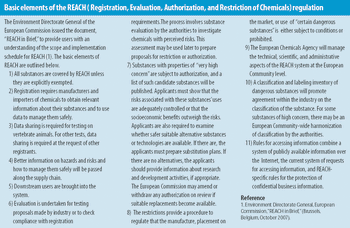
The European Union's REACH initiative has the potential to affect the flow of chemicals into the pharmaceutical suppy chain.

Show blasts off this month in Philadelphia with more suppliers, new trends, and real-world solutions.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration released a final guidance on protocol for testing sterile products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration has issued generics manufacturer Vintage Pharmaceuticals a warning letter stemming from July through August 2007 that found observations pertaining to microbial contamination and failures in testing and documentation procedures.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Biocon will acquire 70% of AxiCorp, ARIAD Pharmaceuticals promoted Richard W. Pascoe to the new position of COO, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Baxter Healthcare Corporation is providing an update to its January 2008 heparin sodium injection 1000 units/mL 10 and 30mL multi-dose vial voluntary recall of nine lots, which the company initiated as a precautionary measure due to an increase in reports of adverse reactions that may be associated with the drug.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration is requesting a 5.7% increase in its budget between the current fiscal year, FY 2008, and FY 2009, for a total $2.4 billion budget.