
Comparative-effectiveness analysis aims to promote appropriate pharmaceutical spending.

Comparative-effectiveness analysis aims to promote appropriate pharmaceutical spending.

Customers complaining lead to some serious explaining.

Brief pharmaceutical news items for June 2008.

The author provides an overview of key regulatory issues facing companies seeking to market their biopharmaceutical agents globally.

Risk management, and its benefits for patients, plays a big role at the PDA Annual Meeting.

With rising drug deveopment costs and burdensome clinical trials, Indian-based firms are transferring their research departments to other entities in hopes of saving cash, mitigating risk, and ultimately, buying back the rewards.

Letting the public inside the drug development process may increase their faith in what we do.
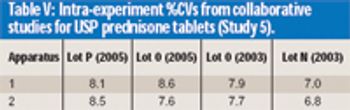
The authors demonstrate that anecdotal reports of prednisone tablet variability are inaccurate.

The good, the bad, and the ugly about direct-to-consumer advertising.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Senate approved a measure (HR 2642) that would provide the US Food and Drug Administration with $275 million in additional funding under a supplemental appropriations bill. The measure now goes before the House.

ePT--the Electronic Newsletter of Pharmaceutical Technology
In a white paper published today, the US Food and Drug Administration described its Sentinel Initiative to create an electronic safety system that tracks drug performance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Federation of Pharmaceutical Industries and Associations (EFPIA), the trade association representing European pharmaceutical manufacturers, issued recommendations to the public consultation launched in March by the European Commission's proposed drug anticounterfeiting measures. EFPIA's proposal includes a ban on drug repackaging.

Equipment and Processing Report
Controlling modular, self-contained process skids (known as plug-and-play components) with an automation system can increase a pharmaceutical manufacturing facility?s flexibility.

Equipment and Processing Report
Manufacturers in the life sciences are increasingly implementing wireless technologies to reduce the overall cost of compliance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Quintiles Transnational to acquire Eidetics, ChemAxon appoints Alex Drijver CEO, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Once again, the US Food and Drug Administration is under fire for not doing its job. This time the issue is direct-to-consumer (DTC) advertising.

ePT--the Electronic Newsletter of Pharmaceutical Technology
To establish user fees for the 2009 fiscal year, the US Food and Drug Administration is asking pharmaceutical manufacturers to provide a list of products and finished-dose manufacturing facilities subject to such fees by June 16, 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Congressional hearings were held last week on the Food and Drug Administration Globalization Act discussion draft.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA removes OAI status for Watson's Florida facility, executive management changes as GSK, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
To keep pace with proposed increases in the number of manufacturing inspections, the US Food and Drug Administration's Office of Regulatory Affairs (ORA) has announced it will increase laboratory capacity and staff.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Merck and Company?s West Point, Pennsylvania facility received a Warning Letter from the US Food and Drug Administration.
Scientists are giving up on a preventive vaccine for AIDS, but there are lessons to be learned.

With counterfeiting on the rise and Europeans worried their backyard is becoming a base for such illegal activity, legislators have proposed a series of solutions that span the continent and abroad.

The authors survey the approved applications of dimethyl sulfoxide USP, PhEur across the healthcare industry and consider the suitability of DMSO from a regulatory and formulation compatibility standpoint.

The US Food and Drug Administration announced its Pharmaceutical GMPs for the 21st Century initiative six years ago. This article reports on the outcome of a recent workshop on this topic and the action plan set forth.