
ePT--the Electronic Newsletter of Pharmaceutical Technology
A group of researchers from Georgia Institute of Technology (Atlanta, GA) are using high-throughput ionization techniques to identify and measure the ingredients in counterfeit drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
A group of researchers from Georgia Institute of Technology (Atlanta, GA) are using high-throughput ionization techniques to identify and measure the ingredients in counterfeit drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Although the number of anti-infective vaccines (as distinct from therapeutic vaccines for cancers and other noninfectious diseases) entering clinical study each year since 2000 has been higher on average than it was in the 1990s, this product area may see little additional growth through the rest of this decade, according to a recentanalysis from the Tufts Center for the Study of Drug Development (Boston, MA).

ePT--the Electronic Newsletter of Pharmaceutical Technology
Late last week, Pliva d.d. (Zagreb, Croatia) announced that the US Food and Drug Administration had granted final approval for its warfarin sodium tablets and azithromycin for oral suspension.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Bayer Group (Leverkusen, Germany) plans to sell the diagnostics division of Bayer HealthCare to Siemens AG (Munich, Germany) for EUR 4.2 billion ($5.36 billion).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration (Rockville, MD) announced that Baxter Healthcare Corp. (Deerfield, IL) signed a consent decree relating to the company's "Colleague" volumetric infusion pump and "Syndeo" patient-controlled analgesic syringe pump.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Vaccine maker Sanofi Pasteur, Inc. received a US Food and Drug Administration Warning Letter, dated June 30, citing "significant deviations" from current good manufacturing practices in the production of monovalent concentrates used in the company?s ?Fluzone? influenza vaccine.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Abbott Labs, Cambrex, Cardinal Health, Charles Ross and Son Co., Janssen, West Pharmaceutical Services

ePT--the Electronic Newsletter of Pharmaceutical Technology
Dutch biotechnology company Crucell NV (Leiden, Netherlands) and its technology partner DSM Biologics BV, a business unit of Royal DSM NV (Heerlen, Netherlands) will open a new research and development center that will specialize on further developing the "PER.C6" human cell line for the expression of recombinant pharmaceutical proteins.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Does the competition from authorized generics really help lower drug prices and boost healthcare savings?
ePT--the Electronic Newsletter of Pharmaceutical Technology
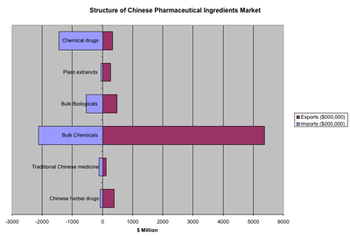
The sixth CPhI China exhibition, presented June 27?29 in Shanghai, offered a showcase for the explosive growth of the Chinese pharmaceutical sector.

University of Buffalo (Buffalo, NY, www.buffalo.edu) researchers have developed a drug delivery system that uses an external magnetic field to guide drug-filled nanocarriers to cultured tumor cells.

ePT--the Electronic Newsletter of Pharmaceutical Technology
MedImmune, Inc. (Gaitherburg, MD) reports that US Food and Drug Administration (Rockville, MD) has approved the company's supplemental biologics license application to use reverse genetics technology to construct new vaccine strains to produce seasonal influenza vaccines.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Biopharmaceutical company Lipoxen PLC (London, UK) has developed a Hepatitis E vaccine using its novel vaccine delivery technology "ImuXen," which the company claims to be easy to manufacture. According to the company, the proprietary liposomal formulation method delivers vaccine materials to the immune system in a manner designed to emulate the response of a natural encounter with the infection agent.

From RFID and pedigrees to PAT and process understanding, industry professionals are eying the growing gap between ambitious high-level guidance and current practice, and wondering how they can bridge this new space and keep in compliance with the untested rules.

A cleverly developed patent portfolio can block or minimize competition long past a patent's 20-year lifespan.

Pharmaceutical Science & Technology News

My audit host clearly knew and lied about the planned changes.

We can learn a lot about today's regulatory environment as a whole if we take it all in.

China is on the rise as a center for pharmaceutical R&D, but companies are still getting their footing for operating in China and the services industry has some maturing to do.

Pharmaceutical Technology Europe
The creation in the Paris Region of a multidisciplinary Institute of Technology will bring together the best researchers in world, innovative SMEs and the research centres of large industrial groups.
Pharmaceutical Technology Europe
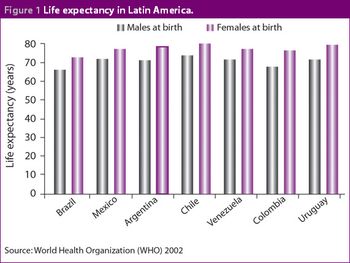
Although it has recently been surpassed by Mexico in terms of value, the Brazilian pharmaceutical market remains of key importance to companies establishing themselves in Latin America. The Brazilian government has focused heavily on improving the healthcare system and this should lead to long-term benefits for its citizens. The Ministry of Health has also attempted to decentralize the management of the healthcare system to more regional and local levels. This has been necessary to account for the different healthcare priorities in different parts of the country.

Last week, the Generic Pharmaceutical Association (GPhA Arlington, VA) praised a proposal by the Senate Agricultural Appropriations Subcommittee that, if approved, would provide $10 million in additional funding for the US Food and Drug Administration?s (Rockville, MD) Office of Generic Drugs.

Baxa Corporation, Eli Lilly, PLIVA, SkyePharma, Wyeth

The US Food and Drug Administration (Rockville, MD) is restructuring its Office of Regulatory Affairs (ORA) into two offices.

The US Food and Drug Administration (Rockville, MD) approved the first generic versions of "Zocor" (simvastatin), the anticholesterol drug by Merck & Co., Inc. (Whitehouse Station, NJ).

On June 15, 2006, the US Food and Drug Evaluation?s Center for Drug Evaluation and Research (Rockville, MD) issued a 7-page warning letter to Ranbaxy Laboratories (Himachal Pradesh, India) for violations to US current good manufacturing practices.

Pfizer, Inc. (New York, NY) reports the United Kingdom's Court of Appeal has upheld the exclusivity of the main patent covering atorvastatin, the active ingredient in "Lipitor."

Johnson & Johnson Company (New Brunswick, NJ) agreed to buy the consumer healthcare business of Pfizer, Inc. (New York, NY) for $16.6 billion in cash.

Coinciding with the 100th anniversary of the US Food and Drug Administration (Rockville, MD), Representative Henry A. Waxman (D-CA) released a statement this week expressing his concern over ?an agency that is adrift and floundering.?

Pfizer, Inc. (New York, NY) will phase out manufacturing operations in Groton, Connecticut, eliminating roughly 300 jobs.