
ePT--the Electronic Newsletter of Pharmaceutical Technology
Drug Sales Up 5.4%...But Growth Rate Slowing

ePT--the Electronic Newsletter of Pharmaceutical Technology
Drug Sales Up 5.4%...But Growth Rate Slowing

ePT--the Electronic Newsletter of Pharmaceutical Technology
Warning Letter: Southern Meds

ePT--the Electronic Newsletter of Pharmaceutical Technology
Cambrex Reports Loss; Advances Biosciences Business

ePT--the Electronic Newsletter of Pharmaceutical Technology
Baxter Wins Contract to Develop Cell-Based H5N1 Vaccine

GPhA Seeks Additional ODG Funding, Not User Fees

ePT--the Electronic Newsletter of Pharmaceutical Technology
Industrial Info Resources reports that North American pharmaceutical and biotechnology companies and life science research institutions are planning a $14.7-billion building boom, with more than 320 active capital projects.

Informex, ORLANDO - The rising influence of custom manufacturers and fine chemicals suppliers from India and China in the global market was evident at Informex 2006, the trade show of custom and batch manufacturers, which is being this week in Orlando. An increase in the number of exhibitors from India and China, expansion plans by several large players, and partnering strategies of Western fine chemical companies, all point to the growing participation of India and China in the fine chemicals market.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Informex, ORLANDO - Facing growing competition from offshore suppliers, several mid-sized custom manufacturers announced expansion plans in specialized technologies at Informex this week.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Informex, ORLANDO - Saltigo GmbH (Leverkusen, Germany, www.saltigo.com), newly formed from the fine chemicals business unit of Lanxess AG (Leverkusen, Germany, www.lanxess.com), made its debut at Informex in Orlando this week. Saltgio will officially launch as a new company in April. It will focus on custom manufacturing and be divided into three principal business lines: pharmaceuticals, specialties, and agrochemicals.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Cambrex (East Rutherford, NJ) to produce Geron (Menlo Park, CA) telomerase anti-cancer vaccine. Generex (Toronto, ON.) files IND for synthetic vaccine to stimulate cell-mediated immunity to avian influenza. Dow Agrosciences (Indianapolis, IN) wins USDA approval for veterinary vaccine, the first manufactured via plant cell culture. G-8 nations pledge billons for vaccine production.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Pharmaceutical Industry has been slow in adopting radio frequency technology (RFID) to help control diversion and counterfeiting, according to a recent study by ABI Research (Oyster Bay, NY, www.abiresearch.com). In fact, only 10 drug products are expected to be shipped with RFID tags or smart chips embedded in the labeling in the coming year.

Informex, ORLANDO - Optimizing a synthesis for an active pharmaceutical ingredient (API) or pharma intermediate is a critical part of the pharmaceutical manufacturing process, and several exhibitors at this year’s Informex highlighted their recent advances in catalysis, biocatalysis, and chiral chemistry. Among the developments: Reaxa to launch biotech affiliate. ChiralQuest develops new chiral ligands. BioCatalytics launches new line. Codexis expands directed evolution biocatalysis program with BMS. Chemtura targets organometallic catalysts for pharma manufacturing.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Securities and Exchange Commission (SEC) has ended its investigation of whether Chiron Corporation (Emeryville, CA, www.chiron.com) violated federal security laws in connection with the previous suspension of Chiron’s license to manufacture “Fluvirin” influenza virus vaccine by the UK Medicines and Healthcare Products Regulatory Agency.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration’s $1.95-billion budget request for the 2007 fiscal increases spending on high-priority programs by $70 million, and cuts support for some existing programs by $52 million.

Akzo Nobel NV (Arnhem, the Netherlands, www.akzonobel.com) plans to spin off its pharmaceutical division by creating a separate company, Organon Biosciences.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Merck wins approval for rotavirus vaccine as Sanofi ships investigational H5N1 vaccine to NIH and CDC explores new flue diagnostics and novel vaccine-production technology.

ePT--the Electronic Newsletter of Pharmaceutical Technology
ASTM International’s (West Conshohocken, PA, www.astm.org) Committee E55 on Pharmaceutical Application of Process Analytical Technology has established a subcommittee “E55.03 on General Pharmaceutical Standards,” to focus on issues relating to quality within a chemistry, manufacturing, and control framework.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Cambrex Corporation (East Rutherford, NJ, www.cambrex.com), a contract manufacturer and provider of life sciences products and services, has retained Bear Sterns & Co Inc. as an advisor as it considers strategic alternatives for the company.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Dow AgroSciences Receives Regulatory Approval for Plant-Made Vaccine

ePT--the Electronic Newsletter of Pharmaceutical Technology
Albany Molecular Research To Acquire ComGenex

FDA is opening the door to continuous learning in process development.

"It was the late 1970s," reports our GMP-Agent-in-Place, "and we used a primitive desktop computer with built-in teletype for our quality control work.

I rarely talk science on the weekends. I spend a good deal of that free time with friends from college performance groups. The conversation centers more on choreography and piano concerti than on CGMPs and chemical reactions. So it always throws me for a loop when these theater and music buffs drop pharmaceutical news into casual conversation.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Lonza Plans Biologics Manufacturing Expansion
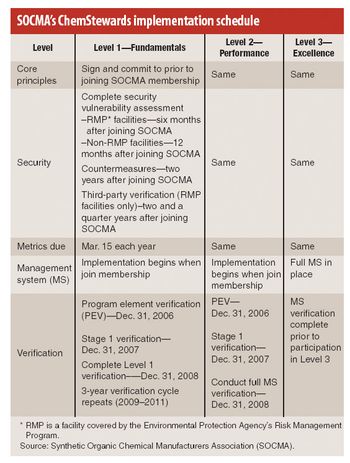
This is a year of change for the Synthetic Organic Chemical Manufacturers Association (SOCMA), the Washington, DC-based trade association representing chemical batch and custom manufacturers. Following the sale of Informex, its flagship trade show, last fall, the association is advancing key programs, most notably its new ChemStewards program, an environmental, health, safety, and security initiative (EHS&S) that its members began implementing last month.

Enterprise information integration delivers needed real-time capability to operational business intelligence.

ePT--the Electronic Newsletter of Pharmaceutical Technology
SAFC Launches SAFC Supply Solutions

Eggs from Transgenic Hens Express Interferon Beta-1a Protein

Preliminary results from a clinical trial of Sanofi Pasteur's (Lyon, France, www.sanofi.com) H5N1 prepandemic influenza vaccine indicate the vaccine is safe and was well-tolerated in 300 healthy volunteers. This study is the first trial of an H5N1 prepandemic influenza vaccine candidate that compared vaccines with and without adjuvants.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Cell-Grown Vaccine Protects Against Avian Flu Virus