
Regulation: FDA Steps Up Action Against Counterfeit Drugs, Warns US Consumers Against Fake Medicine from Mexico

Regulation: FDA Steps Up Action Against Counterfeit Drugs, Warns US Consumers Against Fake Medicine from Mexico

A myopic focus on short-term growth can endanger the community. The question is, can we enlighten our self-interest before the edifice falls down around our ears?

Once considered mainly an afterthought in a company's lifecycle-management strategy, controlled-release dosage forms are now positioned at the forefront of many formulation strategies. In contrast to drug discovery, formulation work focuses not only on the intricacies of the active pharmaceutical ingredient (API), but also on fine-tuning the excipients, the release profile, and the delivery mechanism to provide optimal therapeutic benefit. Because of their wide range of applications and functionalities, especially in controlled-release therapies, polymers are among the most widely used excipients.

In his Feb. 2005 viewpoint article, "In Defense of Singlet Testing," Torbeck (1) draws an important philosophical distinction between "standards" and "specifications." He argues that specifications are criteria selected by manufacturers for statistical control of their products, whereas compendial standards are absolute requirements. This distinction is entirely compatible with modern concepts of statistical process control.

Purdue?s RFID Pedigree Program Enters Pilot Phase

Pharmaceutical Technology Europe
Detection and identification of different polymorphic forms is, therefore, important throughout the drug development and manufacturing process.
Pharmaceutical Technology Europe
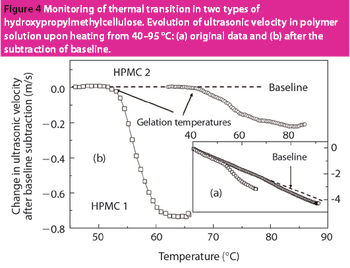
HR-US is a nondestructive technique with enormous potential for the analysis of materials and formulations used in the pharmaceutical industry.

Pharmaceutical Technology Europe
Possible cross-contamination issues should be eliminated at the early stage of the project. The project sponsor should ensure that all relevant personnel from the production, quality control, logistics, and maintenance departments, as well as engineering, are involved in the conceptual stages of a design.
Pharmaceutical Technology Europe
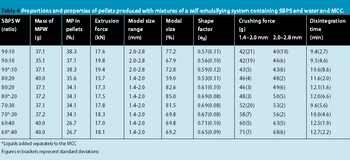
This high quality of pellet roundness is surprising in view of the high extrusion forces required to extrude the wet masses.

Compounding Pharmacies' Lawsuit Against FDA Will Continue

Bedford Recall

GSK and FDA Agree on Consent Decree

Pharmaceutical Technology has joined with INTERPHEX to found GenerationNext, an award program to recognize emerging leaders in pharmaceutical science and technology.

Pharmaceutical science and technology news.

A ready-to-fill closed vial can improve aseptic filling quality and reduce process complexity.

Pharmaceutical Technology Europe
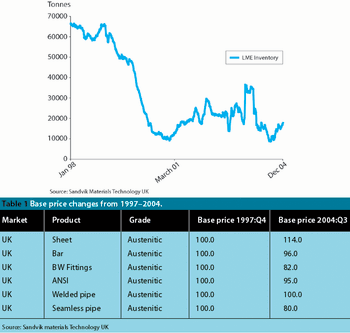
While Europe's political leaders were paying lip-service to creating a more competitive Europe at their spring summit in Brussels, European pharmaceutical technologists were tackling the serious business of turning political rhetoric into practical reality.
Pharmaceutical Technology Europe
Improved products and processes that are cleaner, less expensive, more manageable and take up less space are benefits that you would expect to be snapped up by any industry. But not so in pharmaceutical manufacturing, where traditional methods prevail.

Pharmaceutical Technology Europe
To help reduce the risks of operator exposure and process cross contamination of powdered materials in the pharmaceutical manufacturing environment, the International Society of Pharmaceutical Engineering (ISPE) has published ISPE Good Practice Guide: Assessing the Particulate Containment Performance of Pharmaceutical Equipment.

Pfizer Recalls Lot of Neurontin

Drug Recall

Warning Letter: Compounding at Palace Pharmacy

Pharmaceutical Science and Technology News

The C-SOC team must develop a network of committed industrial partners who will participate in the direction, execution, and evaluation of research and educational activities.

What the industry needs most is not reduced oversight. What we need is, competent, constructive, informed, consistent, fair, and predictable inspection.

The authors argue that chlorine dioxide (CD) is a safe and effective decontaminating agent that can be used for challenging applications.The effectiveness of CD gas for sterilizing complex isolator systems is studied.

At its 2005 Convention, USP passed 13 resolutions that will help define the organization’s path for the next five years and represent the interests of its diverse constituency.

FDA Finalizes Guidances on Minimizing Drug Risk and Submitting Pharmacogenomic Data

FDA New Drug Review Times Continue to Drop in 2004

USP to Publish Spanish Translation of USP-NF; Council of Experts Named