
Enterprise quality management systems can help shift the quality emphasis from corrective to preventive actions.

Enterprise quality management systems can help shift the quality emphasis from corrective to preventive actions.

PTSM: Pharmaceutical Technology Sourcing and Management
Supply chain security and quality group Rx-360 driven by patient safety.

PTSM: Pharmaceutical Technology Sourcing and Management
The European Union takes the lead in a global pilot project on the marketing approval of generic medicines, highlighting the challenges of achieving consensus among different nations.

PTSM: Pharmaceutical Technology Sourcing and Management
The Brazilian economy is being impacted by internal and external factors, and forecast figures are being quickly revised downwards.

Gerresheimer, a company with expertise in pharmaceutical and medical technology, announced that it is applying an integrated mold qualification and validation concept for a fast and cost-efficient time-to-market. The company notes that this approach provides process windows for reliable large-scale production and offers options for different deployment scenarios and qualification levels.

A customer complaint prompts the recall of a lot of anticoagulants from a plant in Puerto Rico.

Contract Packaging Resources issued a voluntary recall of naproxen sodium tablets because some cartons contain bottles of ibuprofen.

The agency recommends that companies developing drugs to treat Ebola apply for orphan drug designation.

A selection of articles and news about quality metrics.

After almost two years of anticipation, Janet Woodcock, director of the Center for Drug Evaluation and Research (CDER), has administration approval for organizational changes to bolster programs and policies to ensure drug quality.

Hospira voluntary recalls certain lots of several LifeCare products due to potential for leakage.

Iclusig?s original indication will remain unchanged after a thorough examination of its safety information by EMA.

The European Medicines Agency releases highlights from its Pharmacovigilance Risk Assessment Committee safety review meeting in October.

FDA outlines procedures for having a Critical Path Innovation Meeting with CDER.

FDA draft guidance outlines ways to minimize risk from acetaminophen-containing over-the-counter pediatric liquid drug products.

We?ve just opened new pharmaceutical laboratories! Our experienced scientists offer independent chemical and microbiological quality control testing of APIs, raw materials, finished products and medical devices in compliance with cGMP

FDA releases question and answer draft guidance on drug product tracing and licensing requirements.

Impurities, particulates, and a lack of communication are among Hospira's recent violations described in a warning letter from FDA.

Demand for new therapies and vaccines spotlights production challenges.

The recovery of an occasional mold does not merit any particular concern. On the other hand, evidence of mold proliferation indicative of infection of facilities or equipment must be taken seriously and requires the prompt implementation of corrective and preventive actions.

Regulatory agencies in Europe are working to harmonize the marketing approval pathway of generic medicines.
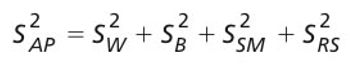
A risk-based guard band surrounds a specification limit and is derived from the uncertainty of the reportable value of the analytical procedure, which includes the uncertainty in the reference standard. The author discusses requirements for generating a reportable value and calculating the associated measurement uncertainty.

Brazil's pharmaceutical industry is optimistic, but is the pharmaceutical market growing steadily or showing signs of instability?

Siegfried Schmitt, Principal Consultant, PAREXEL International, discusses the benefits of automated processes.

PTSM: Pharmaceutical Technology Sourcing and Management
Multidirectional collaboration is critical for the new pharma business model; cloud-based information services can offer a communications alternative.