
ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued a draft guidance, Contents of a Complete Submission for the Evaluation of Proprietary Names, on Nov. 24, 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued a draft guidance, Contents of a Complete Submission for the Evaluation of Proprietary Names, on Nov. 24, 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
In a letter to Acting Comptroller General Gene Dodaro dated November 19, 2008, Congressman Joe Barton (R-Texas) requested that the US Government Accountability Office (GAO) review the US Food and Drug Administration's handling of the highly-publicized, tainted heparin scare that occurred in 2007 and 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Ranbaxy Laboratories (Gurgaon, Haryana, India) responded to an investigation of violations at two of its manufacturing plants, according to a Reuters Health report.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Although the number of annual new-drug approvals in the United States has steadily declined during the past ten years, pharmaceutical companies still prefer to introduce new products in the US first, according to a report published by the Tufts Center for the Study of Drug Development.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug issued a draft guidance on Tuesday, Nov. 18, titled Process Validation: General Principles and Practices for comment. The guidance is meant to serve as a revision to the 1987 Guideline on General Principles of Process Validation.

ePT--the Electronic Newsletter of Pharmaceutical Technology
US marshals seized 11 lots of heparin from Celsus Laboratories (Cincinnati, OH) at the request of the US Food and Drug Administration.

ePT--the Electronic Newsletter of Pharmaceutical Technology
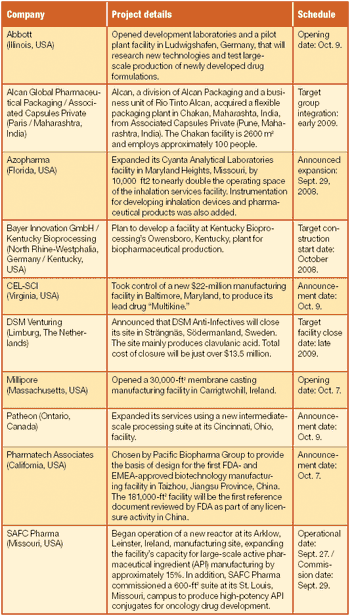
Also, Patheon opens new European headquarters, Cynvec appoints Frank D. Stonebanks president and CEO, more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Steering Committee and Expert Working Groups of the International Conference on Harmonization are meeting this week in Brussels to discuss current harmonization efforts.

Equipment and Processing Report
PAT may reduce costs by helping companies control process variability, improve yields, reduce waste, and produce high-quality therapies consistently. Companies that have not yet embraced PAT may find its potential to reduce expenses a compelling argument in its favor during this time of financial difficulty.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency's (EMEA) Committee for Medicinal Products for Human Use recommended that the status of GlaxoSmithKline's anti-obesity drug "Alli" (orlistat) be switched from prescription-only to nonprescription.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration sent Warning Letters to Bayer HealthCare (Morristown, NJ) about the company's "Bayer Women?s Low Dose Aspirin + Calcium" and "Bayer Aspirin with Heart Advantage" over-the-counter (OTC) products.

This position paper describes a model for the future that would provide appropriate standardization, facilitate drug registration and support regulatory agencies.

Policymakers weigh new rules to ensure the safety and quality of drugs made with tiny particles.

Attendees at a recent workshop endorsed new methods to detect metals in drugs, dietary supplements, and food ingredients.

The Indian government may soon monopolize its pharmaceutical industry to cut costs and improve healthcare, but the move is sounding off alarm bells with the companies whose drug products are under review.

The US Food and Drug Administration seeks to understand nanotechnology better and exercise appropriate oversight over products that incorporate it.

Pharma companies could benefit from the lessons learned in this fall's financial crisis.

Production problems come in all shapes, sizes, and ... species.

IPEC Chairman Dave Schoneker discusses current efforts toward facilitating regulatory reviews of new excipients.


Scott Sutton discusses the current state of USP ‹1117› and USP's plans for future revisions.

Pharmaceutical Technology Europe
During the past two decades, regulations have evolved in both Europe and the US to accommodate the technological developments in the pharmaceutical, biotechnology and medical device industries.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Government Accountability Office (GAO) released a report last week that examines and provides recommendations for the US Food and Drug Administration's foreign inspection process, including the agency's data management, inspection frequency, and oversight of problems identified during inspections.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration has awarded the National Institute for Pharmaceutical Technology and Education (NIPTE), a not-for-profit organization comprising 11 universities, a $1.19 million contract to develop quality by design (QbD) guidance elements for design space and scale-up of unit operations.