
As Europe’s bio/pharma market learns that breaking up is hard to do, it also must address productivity, regulatory, and drug pricing challenges.

As Europe’s bio/pharma market learns that breaking up is hard to do, it also must address productivity, regulatory, and drug pricing challenges.

The agency has published its refuse-to-receive standards guidance for abbreviated new drug applications.
Equipment and Processing Report
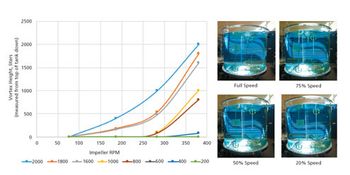
Quantitative and qualitative tools allow better understanding of mixing in a single-use bioprocessing system.

PTSM: Pharmaceutical Technology Sourcing and Management
Pharma Tech introduced a new high-speed bottling line to its facility in Union, MO.

PTSM: Pharmaceutical Technology Sourcing and Management
AGC adds second biopharma contract manufacturer with acquisition of CMC Biologics.

PTSM: Pharmaceutical Technology Sourcing and Management
Novasep’s three facilities located in France and Germany are free of Form 483.

PTSM: Pharmaceutical Technology Sourcing and Management
The company has manufactured double-digit number of batches on the new filling line, for use in early-stage clinical trials.

PTSM: Pharmaceutical Technology Sourcing and Management
The companies entered into a manufacturing agreement for voclosporin active pharmaceutical ingredient.

PTSM: Pharmaceutical Technology Sourcing and Management
The two companies have entered into an agreement to provide Suzeken with access to AmerisourceBergen’s Cubixx inventory solution.

PTSM: Pharmaceutical Technology Sourcing and Management
The $2-billion facility will be located in Clayton, North Carolina.

The contract service provider entered into a contract agreement with the US Army to provide services in the development of treatments of malaria.

Portola signed a $50-million loan agreement with BMS and Pfizer to provide additional funding for the development of andexanet alfa.

Baxter will acquire Claris Injectables Limited, adding additional generic injectable capabilities to the company’s porfolio.

The agency put Baoying County Fukang Medical Appliance Co., Ltd. on import alert after observing violations at the company’s Yangzhou City facility.

A pilot project, beginning in 2017, will support the development of biosimilars.

The companies entered a manufacturing agreement for the future commercial production of Lenti-D and LentiGlobin product candidates.

Emergent signed a follow-up contract to provide 29.4 million doses of BioThrax to the Strategic National Stockpile.

The agency announces that 81 medicines overall were recommended in 2016.

Univercells received a grant from the Bill & Melinda Gates Foundation for the development of a vaccine manufacturing platform.

PTSM: Pharmaceutical Technology Sourcing and Management
Lonza adds CDMO, capsule, and consumer healthcare and nutrition capabilities with $5.5-billion acquisition of Capsugel.

The commission concluded its P4Bio pilot phase with the adoption of the monograph for etanercept.

PDA is conducting a survey of industry members challenged by the complexity of making post-approval changes.

FDA’s Center for Drug Evaluation and Research makes plans for implementation of the 21st Century Cures Act that include patient-focused drug development.

Oxford Genetics received £1.61 million from Innovate UK to explore computational and synthetic biology approaches for optimized mammalian bioproduction.

IPEC-Americas held elections for executive committee positions at the annual board of trustee’s dinner.

GlaxoSmithKline opened a new vaccines R&D center in Rockville, MD creating up to 200 new jobs.

PTSM: Pharmaceutical Technology Sourcing and Management
DuPont received a grant from the Bill & Melinda Gates Foundation to produce pharmaceutical proteins.

BMS changes its US geographic footprint with R&D investments and closures.

PTSM: Pharmaceutical Technology Sourcing and Management
Saneca Pharma has received a EUR1.5 million grant from the Slovak Ministry of Education, Science, Research, and Sport, which will be used to drive forward new R&D initiatives for API manufacturing.

Congress enacted the 21st Century Cures legislation, which shores FDA operations and supports biomedical research at the National Institutes of Health.