
At the American Society of Hematology Annual Meeting, Novartis and Kite Pharma both released results from early-phase clinical trials with CAR-T therapies.

At the American Society of Hematology Annual Meeting, Novartis and Kite Pharma both released results from early-phase clinical trials with CAR-T therapies.

Spark Therapeutics and Selecta entered a license agreement for Selecta’s gene-therapy co-administration technology.

FDA issued a warning letter to Interquim, SA for CGMP deviations at its Barcelona API facility.

Takeda and Lightstone have launched Cerevance, a neuroscience company focused on developing therapeutics for neurological and psychiatric disorders.

JHL Biotech and Sanofi collaborate on the development and commercialization of biological therapeutics in China.

Through a license agreement with the Broad Institute, Charles River Laboratories will now be offering gene-editing engineering technology.

QuintilesIMS forecasts global spending on medicine will reach $1.5 trillion by 2021.

Dara Corrigan examines the Mutual Reliance Initiative as a method for expanding FDA’s inspection capabilities in Europe and beyond.

Researchers test the efficacy of a new polymer that is an alternative to PEG for drugs used to treat type 2 diabetes.

FDA and BARDA awarded a contract to Continuus Pharmaceuticals to develop an end-to-end continuous manufacturing process for solid-dosage drugs.

Susan Schniepp, distinguished fellow at Regulatory Compliance Associates, discusses training personnel on a limited budget.

Boehringer Ingelheim and China Southeast University announced a joint research collaboration to develop new regenerative treatments for hearing loss.

The UK’s NICE recommended pembrolizumab for the treatment of patients with advanced lung cancer, reversing the institute’s earlier decision.

PTSM: Pharmaceutical Technology Sourcing and Management
Recipharm and Laccure AB signed a commercial collaboration for the manufacture and delivery of Laccure’s bacterial vaginosis treatment.

The company opened a new facility in Dublin for its cell performance technology.

PTSM: Pharmaceutical Technology Sourcing and Management
Vetter’s Ravensburg data processing center received certification from a German industrial testing organization, certifying that the company’s customer and process data are protected.

WMFTG acquired Aflex Hose adding polytetrafluoroethylene-lined flexible hoses to its fluid path product portfolio.

AstraZeneca entered a collaboration with Bicycle Therapeutics for the identification and development of bicyclic peptides.
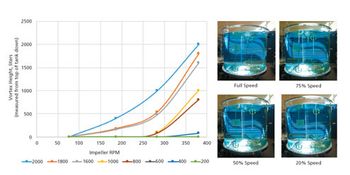
Quantitative and qualitative tools allow better understanding of mixing in a single-use bioprocessing system.

Rookie API developers beat pharma at its own game.

Europe-based bio/pharma employees are unified on satisfaction with employment conditions-and dissatisfaction with salaries.

The House-approved 21st Century Cures legislation supports initiatives for medical research, drug development, and faster approvals, but comes up short on actual appropriations.

PTSM: Pharmaceutical Technology Sourcing and Management
Idifarma has acquired a Bosch GKF-702 capsule filling machine that can manufacture 3000 to 42,000 capsules per hour.

Takeda will invest more than 100 million Euros to build a new manufacturing plant for its dengue vaccine candidate in Singen, Germany.

Agilent’s new facility in Folsom, California includes laboratory, order fulfilment, and warehousing space.

Mylan signed a sub-license agreement with the Medicines Patent Pool to develop a generic version of daclatasvir to market in developing countries.

The commission approved future plans, appointed members, and adopted texts during its November 2016 session.

PTSM: Pharmaceutical Technology Sourcing and Management
Catalent adds two softgel facilities and packaging capabilities with acquisition of Canada-based Accucaps.

PTSM: Pharmaceutical Technology Sourcing and Management
Patheon adds API manufacturing capacity with acquisition of Roche’s Florence, SC facility.

FDA has revised its draft guidance to provide greater clarity and focus. This version would establish an initial voluntary phase to allow regulators and industry to focus, first, on a limited set of metrics.