
The company is opening two offices in the United States that will offer serialization, automation, and process control services.

The company is opening two offices in the United States that will offer serialization, automation, and process control services.

The company is voluntarily recalling one lot of Oxacillin for Injection, USP, 10 g.

PTSM: Pharmaceutical Technology Sourcing and Management
CDMO Alcami adds HPAPI capacity and cryogenic capabilities to its Wisconsin facility.

PTSM: Pharmaceutical Technology Sourcing and Management
Fresenius Kabi will add to its generic, sterile injectable manufacturing at its Melrose Park, Illinois site.

Arbor Pharmaceuticals is voluntarily recalling Cetylev (acetylcysteine) effervescent tablets due to inadequate seal of the blister pack.

TOPAS Advanced Polymers announces its COC materials are compliant with new USP standard for pharma plastic packaging systems.
Equipment and Processing Report
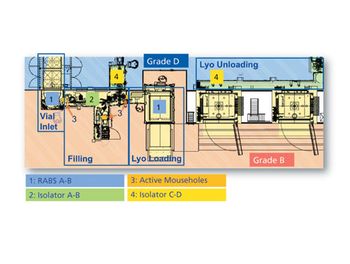
The design of Baxter BioPharma Solutions’ aseptic filling lines provides a case study in customizing containment systems for multi-product lines

PTSM: Pharmaceutical Technology Sourcing and Management
The Chinese facility was cited for data integrity violations.

PTSM: Pharmaceutical Technology Sourcing and Management
The Group is focusing on standardizing data exchanges between the enterprise serialization management function and product packaging lines.

Nine public health organizations submitted a letter to US House and Senate committees citing concerns about Section 11 of the FDA and NIH Workforce Authorities Modernization Act.

The bio-incubator, located at the James Cook University Hospital, will give early-stage biotechnology and life-sciences companies access to facilities to support biomedical research.

Flawed testing and analysis and a failure to analyze the root cause of customer complaints were among the top problems noted in FDA's letter to the Florida-based transdermal drug manufacturer.

PTSM: Pharmaceutical Technology Sourcing and Management
The agency publishes guidance on the appropriate classification of co-crystal solid-state forms.

The company received breakthrough therapy designation for esketamine, a treatment for major depressive disorder with imminent risk for suicide.

PTSM: Pharmaceutical Technology Sourcing and Management
Piramal Enterprises has entered into an agreement to acquire 100% stake in Ash Stevens all by cash for a consideration of $42.95 million plus an earn-out consideration capped at $10 million.

Irvine Scientific’s new product range includes chemically-defined, serum-free media, to increase productivity of viral vectors and recombinant proteins in suspension cultures.

A KPMG survey reveals that approximately 84% of pharma and medical device executives plan to add jobs in next 12 months.

PTSM: Pharmaceutical Technology Sourcing and Management
Agilent Technologies announces plans to build a new oligo manufacturing facility in Colorado that will double current capacity.

CPhI Pharma Awards’ panel doubles in size and welcomes experts from around the world.

Mallinckrodt announced a planned merger agreement with Stratatech Corporation, a regenerative medicine company.

The agency has adopted guidelines on the pharmacovigilance of biological drugs.

A new study reveals a potential new approach to treat sickle cell disease and beta thalassemia using CRISPR-Cas9 gene-editing technology.

Jacobs Engineering will provide engineering and construction services to expand the Novartis site in Huningue, France.

The EDA offered Allergan tax credits in order to encourage the company to remain in the state.

PTSM: Pharmaceutical Technology Sourcing and Management
The GMP-certified facility will produce TruTag microtags for global pharmaceutical customers.

Quebec, Canada-based contract development and manufacturing organization has joined the Pharma & Biopharma Outsourcing Association.

Zika vaccine development is hindered by technical challenges and funding shortfalls.

Pearl River had set the scene for vaccine breakthroughs, cGMP challenges, and ambitious Lean and operational excellence programs

The work will focus on addressing a key bottleneck to progress: he complexity and instability of HIV envelope proteins, which has limited the amount of material that can be evaluated in clinical trials.

Researchers at the ARC Centre of Excellence in Convergent Bio-Nano Science (CBNS) in Melbourne, Australia have developed a drug-delivery technology that circumvents first pas metabolism in the liver, thereby, enabling higher drug levels to reach the systemic circulation.