
ePT--the Electronic Newsletter of Pharmaceutical Technology
Zydus Cadila, a pharmaceutical company based in Ahmedabad, Gujarat, India, will begin conducting trials for a H1N1 vaccine.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Zydus Cadila, a pharmaceutical company based in Ahmedabad, Gujarat, India, will begin conducting trials for a H1N1 vaccine.

ePT--the Electronic Newsletter of Pharmaceutical Technology
On Jan. 4, 2010, Novartis (Basel) and Nestlé began completing their 2008 agreement, under which Novartis will acquire Nestlé's remaining 52% stake in eye-care company Alcon (Hünenberg, Switzerland).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration released a warning to the public on Dec. 29, 2009 about criminals posing as FDA special agents and other law enforcement personnel as part of an international extortion scam.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Bioject Medical Technologies establishes alliance with MPI Research; Biogen Idec CEO to retire; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Microelectronics offers "striking opportunities" for advancing biomedical research and creating new markets for the medical sciences industry.
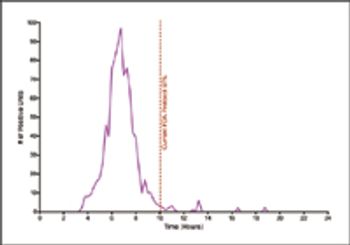
This study used biological indicators containing Geobacillus stearothermophilus spores and a new technology to continuously monitor incubated BIs and record nonsterile results.

A comprehensive book about mass transfer benefits from the author's personal touch.
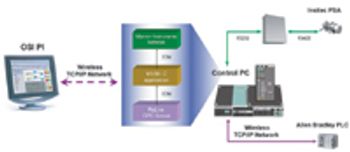
The authors describe the implementation of an on-line particle-size analyzer on an active pharmaceutical ingredient milling operation at a commercial site.
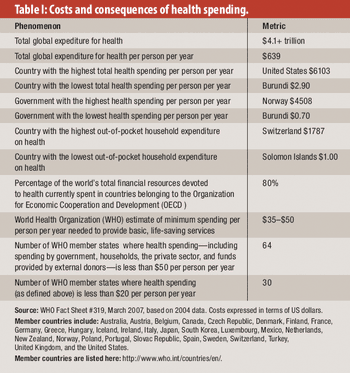
From healthcare to corruption to life expectancy, here's what we can learn from the past decade.

Directors and staff miss the mark when it comes to following procedures.

ePT--the Electronic Newsletter of Pharmaceutical Technology
BioNanomatrix (Philadelphia) names Edward Erickson president and CEO; and More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Hospira, a specialty pharmaceutical company, has agreed to acquire Orchid Chemicals' Pharmaceuticals for $400 million.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Merck & Co. Acquires Avecia Biologics; Ambrilia Biopharma Closes Manufacturing Facility; And More.

Pharmaceutical Technology Europe
Fraud and abuse in healthcare costs individual governments as much as $23 billion a year, according to estimates from the World Health Organization (WHO).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Public Health Alliance published its comments about the Falsified Medicines Directive currently under debate by the European Parliament and Council.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Mergers and acquisitions will contribute almost two-thirds of peer-set sales growth until 2014, according to market analyst Datamonitor.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Eli Lilly outlined its growth strategy at its annual meeting with the investment community last week.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Abbott to acquire Starlims Technologies; GSK Biologicals to form alliance with Intercell.

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment & Processing Report, reviews the Editor's Picks for the December 2009 edition from Sepha and AdvantaPure.

Pharmaceutical Technology Europe
Do advanced therapies represent medical miracles or risky business? This is the question that is debated by an Informa analyst in an Executive Briefing for Scrip.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency unveiled several changes to the organization, including a new structure and visual identity.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: Also, Pfizer and Protalix enter agreement; Watson acquires Arrow Group; and more.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Congressional Budget Office released a report that analyzes the pharmaceutical industry?s advertising strategies.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Personalized medicine has been described by analysts PricewaterhouseCoopers as a "disruptive influence" that will create both opportunities and challenges for traditional healthcare.

Pharmaceutical Technology Europe
The private industry has been urged to join a public sector commitment fund in the UK to advance manufacturing R&D in bioprocessing, following the investment of approximately £9 million from two UK research councils.

Pharmaceutical Technology Europe
European business leaders are missing opportunities to reduce their environmental footprint and drive efficiencies through document governance, according to research from Ricoh, an electronic solutions provider.

ePT--the Electronic Newsletter of Pharmaceutical Technology
sanofi-aventis (Paris) announced last week that it has signed a memorandum of understanding (MoU)with Prominvest.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Novartis officially inaugurated its large-scale flu cell-culture vaccine and adjuvant manufacturing facility in Holly Springs, North Carolina.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention posted a proposed revised standard for what should and should not appear on the ferrules and cap overseals of medication vials.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Merck & Co. extends collaboration with Idera Pharmaceuticals; Pfizer establishes R&D center in China; more...