
ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, new European Generic medicines Association president begins term.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, new European Generic medicines Association president begins term.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pharmaceutical Technology talks to FDA's Justina A. Molzon about current discussions with international regulators, and how, after 20 years of harmonization activities and the common technical document, ICH has benefited regulatory authorities.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Guidance has been published in the United Kingdom regarding a new European variations regulation that comes into effect on Jan. 1, 2010.
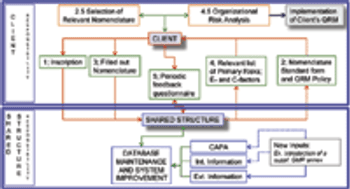
Representatives to the PIC/S provide an example of methodology for implementing ICH Q9.

Readers can learn about the importance of measuring and controlling water activity in a comprehensive new book.

Dumbed-down presentations and poor speaker selections are destroying a valuable industry tool.

With so many healthcare and pharmacy websites, consumers could use the agency's nod of support.

Polymorph screening is a critical step in developing an active pharmaceutical ingredient for a pharmaceutical formulation.

Blame it on cafeteria gossip, outdated procedures, and major miscommunication.
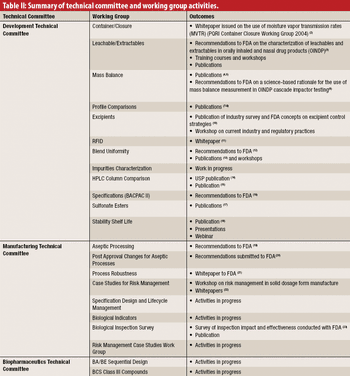
The authors describe the development and organization of the Product Quality Research Institute and highlight some of the important projects conducted by its Working Groups.

Pharmaceutical Technology Europe
It's the end of the year already and I hope you are looking forward to the festive period and the opportunity to wind down and spend some quality time with family and friends.

The end of 2009 is creeping closer and analysts are already looking towards 2010 and wondering what new challenges are in store for the pharma industry.

Pharmaceutical Technology Europe
An agreement between sanofi-aventis and the Medicines for Malaria Venture (MMV) has lead to the initiation of the largest safety and efficacy study of an antimalarial drug.

Pharmaceutical Technology Europe
After further reviewing data on the H1N1 pandemic vaccines approved in Europe, the EMEA has reaffirmed their balance of benefits and risks in the context of the current H1N1 influenza pandemic. Additionally, the World Health Organization (WHO) has issued an update on the H1N1 situation in Europe.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Society for Chemical Manufacturers and Affiliates (SOCMA) provided an update as to its grassroots efforts regarding the recently passed Chemical and Water Security Act of 2009 (HR 2868).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration has issued 22 Warning Letters to website operators as part of its International Internet Week of Action.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: Merck KGaA to build R&D hub in Beijing; Depomed appoints VP of operations; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency confirmed the safety and efficacy of Focetria and Pandermix, and noted that data on Celvapan are still being analyzed.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Five percent of consumers across five European countries suspect they may have received a counterfeit prescription, and 1% believe that they definitely have, according to research conducted by ICM on behalf of patient safety communications company Aegate (UK).

Pharmaceutical Technology Europe
Engineers at a UK university claim to have developed technology that can be used in existing chemical reactors to ensure right first time drug crystal formation, which is crucial to their efficacy and the efficiency of pharmaceutical manufacturers' operations.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Last week, GlaxoSmithKline (GSK, London) agreed to donate 50 million doses of its adjuvanted pandemic influenza A (H1N1) vaccine to the World Health Organization (WHO).

ePT--the Electronic Newsletter of Pharmaceutical Technology
Around half of all multiple sclerosis (MS) patients that are eligible for treatment do not receive it and one in five of those patients that do begin therapy, delay doing so because of fear and anxiety over the treatment process, and not fear and anxiety about the disease.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Following the its $68-billion acquisition of Wyeth (Madison, NJ), Pfizer (New York) Pfizer detailed plans for its global research and development (R&D) network, which includes consolidation of its R&D facilities.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The international pharmaceutical supply-chain consortium Rx-360 held a launch meeting in Europe last week with more than 135 industry and regulatory representatives in attendance.

ePT--the Electronic Newsletter of Pharmaceutical Technology
FDA has approved a fifth influenza A (H1N1) vaccine, manufactured by ID Biomedical.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: Genzyme provides updates on enzyme replacement products and FDA complete response letter; Watson appoints VP of global operations; more...

Equipment and Processing Report
Pigging is a sustainable strategy that can aid in cleaning and product recovery and improve a manufacturing line's efficiency and throughput.

Equipment and Processing Report
Recent advances in microreactor technology are improving the application and scale at which the technology can be applied.

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment & Processing Report, reviews the Editor's Picks for the November 2009 edition from Enercon and Schreiner.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: 3M forms agreement with VaxInnate; TRIN Pharma appoints CEO; More...