
ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer's first updated pipeline since its acquisition of Wyeth includes fewer projects than before and is targeted to specific diseases.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Pfizer's first updated pipeline since its acquisition of Wyeth includes fewer projects than before and is targeted to specific diseases.

ePT--the Electronic Newsletter of Pharmaceutical Technology
FDA Releases Annual Guidance Agenda; OMB Urges Obama to Issue Revised Executive Order.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Less than a week after president Obama proposed a spending freeze on nonsecurity-related federal programs, the US Food and Drug Administration released its fiscal year 2011 budget request, calling for a 23% increase.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Cephalon Buys Mepha; BASi's CEO Retires; and More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
AstraZeneca announced this week that it plans to undertake further restructuring in its research and development operations, resulting in the elimination of 3500 jobs.

Sharing too much-or too little-information can have disastrous onsequences.

FDA impersonators and counterfeit drugs threaten the public's trust in online pharmacies.

A look at the formulation challenges in pancreatic enzyme products.

A recent book provides information about formulating biopharmaceuticals that is easy to swallow.

Regulators and industry move to require inspections of API manufacturing facilities.

When it comes to healthcare rform, we must not overlook investment in innovative technologies.
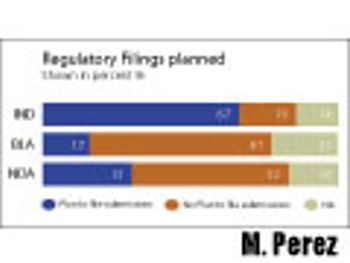
How to cut time and cost by re-using already submitted documents.
We never thought implementing complex changes could become more cumbersome.

Pharmaceutical Technology Europe
With the pharmaceutical industry being in a current state of unrest in light of the recent spate of mergers, acquisitions, job cuts, divestments, patent expiries, and so on, we wanted to know how you felt about all these changes that will have directly or indirectly affected you.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Novartis CEO To Step Down; DSM To Close Netherlands Facility; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
New York Governor Wants Stricter Limits On Pharmaceutical Marketing; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Director-General of the European Generic medicines Association has reiterated the group's Vision 2015 plan.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Two US senators have introduced the Nanotechnology Safety Act of 2010, which would establish a program within the US Food and Drug Administration to assess the health and safety implications of nanotechnology in everyday products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Keiji Fukuda, special adviser on pandemic influenza to the World Health Organization's (WHO) director-general, told a hearing at the Council of Europe that WHO's response to the influenza (H1N1) pandemic was not improperly influenced by the pharmaceutical industry.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Medicines Agency has launched a public consultation on its Road Map to 2015, a strategic vision that lays out the agency's priorities for the next five years.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Lonza Group (Basel) plans to reinforce its presence in Asia, including its platform in Nansha, China, and to close its Conshohocken (Riverside), Pennsylvania; Shawinigan, Canada; and Wokingham, United Kingdom, sites in 2010.

ePT--the Electronic Newsletter of Pharmaceutical Technology
FDA Issues Warning Letter To McNeil Healthcare; Charles River Laboratories Will Suspend Operations At Massachusetts Facility.

ePT--the Electronic Newsletter of Pharmaceutical Technology
FDA Thanks Healthcare Workers For H1N1 Response; And More.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The EU's Commissioner-designate for Health and Consumer Policy, John Dalli, said that he is looking forward to the challenge of integrating the pharmaceutical area into public health as part of his role.

ePT--the Electronic Newsletter of Pharmaceutical Technology
McNeil Consumer Healthcare is voluntarily recalling certain lots of over-the-counter products in the Americas, the United Arab Emirates, and Fiji.

Equipment and Processing Report
Could flexible manufacturing change the standards for biopharmaceutical production? To find out, Equipment and Processing Report talked to James Robinson, vice-president of technical and quality operations at biotechnology company Novavax (Rockville, MD).
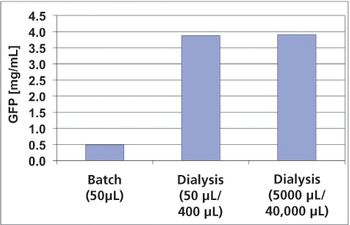
Equipment and Processing Report
The authors sought to improve the productivity of protein synthesis by using a highly active cell-free extract from Escherichia coli and by optimizing buffer conditions and shaking conditions.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The United States Pharmacopeial Convention is recalling USP 33?NF 28.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Many life-sciences companies are likely to execute an acquisition in the next 12-24 months, according to a survey by Ernst & Young.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Commission has asked certain pharmaceutical companies to submit copies of their patent-settlement agreements.