
Contract research organizations such as Covance are heading further east through Europe.

Contract research organizations such as Covance are heading further east through Europe.
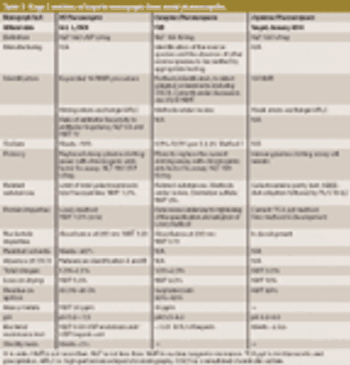
USP workshop participants support new methods to safeguard heparin products but desire international harmonization. This article contains bonus online-exclusive material.

Pharmaceutical Technology Europe PTE
The EMEA intends to fully implement its Product Information Management (PIM) approach, an initiative designed to improve information management, into the centralized procedure within 2 years, according to a statement of intent from the agency.
A review of NIPTE's core projects and its plans for training-and retraining-the pharmaceutical industry.

This article introduces the "Q.U.E.S.T." approach for vendor qualification, a practical and compliant methodology for pharmaceutical and biopharmaceutical companies to qualify vendors and hence make well-informed purchasing-related decisions.

As the pharmaceutical industry moves further into Central and Eastern Europe and the Commonwealth of Independent States, several standard-setting and regulatory bodies are also increasing collaboration in the region, particularly in Russia.

The author describes the framework needed to implement QbD and achieve the deeper process understanding that is fundamental to QbD.

This article is part of a Special Report on the Emerging Markets of The East, October 2009

The author analyzes, from an agency perspective, whether question-based review has improved product quality or made the review process easier for regulators or for industry.

Pharmaceutical Technology Europe
Well, the summer's well and truly behind us and Madrid beckons for this year's CPhI Worldwide.

Pharmaceutical Technology Europe PTE
How manufacturers can accelerate cashflow by cutting out the paper trail.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA bans candy and fruit-flavored cigarettes, EMEA moves to improve information management.

ePT--the Electronic Newsletter of Pharmaceutical Technology
A new report evaluating the biotechnology sector in new European Union member states and candidate countries has described biotech developments as "uneven" because of contrasting economic strategies in different countries.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The biopharmaceutical company Seattle Genetics (Bothell, WA) introduced a sugar-engineered antibody (SEA) technology that is designed to increase the potency of monoclonal antibodies through enhanced effector function.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued a proposed rule to clarify the current good manufacturing practice (CGMP) requirements applicable to combination products in the Sept. 23 Federal Register.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: Boehringer Ingelheim will acquire Wyeth's animal health business; Amsterdam Molecular Therapeutics appoints CEO; more...

Pharmaceutical Technology Europe PTE
Rene Kummer of Sigpack Systems speaks about the company's new pharma toploader, which features an innovative box transportation unit.

ePT--the Electronic Newsletter of Pharmaceutical Technology
On September 25, the European Medicines Agency's (EMEA) Committee for Medicinal Products for Human Use (CHMP) recommended the authorization of two vaccines for use in Europe against the H1N1 influenza: GlaxoSmithKline's (GSK) Pandemrix and Novartis's Focetria.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Abbott Laboratories (Abbott Park, IL) agreed to acquire the pharmaceutical business of the Solvay Group (Brussels, Belgium) for EUR 4.5 billion ($6.6 billion) in cash, according to an Abbott press release.

Pharmaceutical Technology Europe PTE
Vetter (Germany) is intending to open a technologically advanced customer services facility at the end of 2009 in Chicago (US).

Pharmaceutical Technology Europe PTE
The Gerresheimer Group is opening two new centres: an R&D center for medical plastic systems in the US and a new production center for pharmaceutical plastic packaging in Spain.

Pharmaceutical Technology Europe PTE
A joint venture between a charity and a pharma giant has led to the creation of the Hilleman Laboratories, which will use a not-for-profit operating model to develop and deliver vaccines to low-income countries.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration plans to hold a public hearing to discuss the promotion of agency-regulated drugs and medical devices on the Internet and using social media tools, according to a notice in the Sept. 21, 2009, Federal Register.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA extended the deadline for the pilot program for the submission of CMC information.

ePT--the Electronic Newsletter of Pharmaceutical Technology
A report from the European Medicines Agency (EMEA) has highlighted the critical fact that there are not enough new antibiotics in the pharmaceutical industry's pipeline to fight multidrug-resistant bacteria.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Many pharmaceutical companies in the United Kingdom are turning to basic business-skills training to help them emerge from the recession stronger than before.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: Wyeth and Ambrx form development pact; Elite Pharma appoints CEO and CSO; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration sent Bayer Schering Pharma (Berlin) a Warning Letter on Aug. 5, 2009, citing deviations from current good manufacturing practice in the manufacture of nonsterile active pharmaceutical ingredients (APIs).

Pharmaceutical Technology Europe PTE
The current pharmaceutical model is in danger of becoming "irrelevant in the context of 21st century's global healthcare needs", according to a white paper published by global management consultancy firm A.T. Kearney.

Pharmaceutical Technology Europe PTE
AM Technology (UK) has joined an EU-funded project that seeks to develop flow processes for the industrial synthesis of pharmaceutical compounds.