
ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Crucell and DSM announce deals with GSK, Talecris, and CSL; Nobel Prize winner Luc Montagnier joins Viral Genetics; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Crucell and DSM announce deals with GSK, Talecris, and CSL; Nobel Prize winner Luc Montagnier joins Viral Genetics; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration issued a draft guidance, Genotoxic and Carcinogenic Impurities in Drug Substances and Products: Recommended Approaches.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration dedicated historic Building One at the White Oak Federal Research Center in Silver Spring, Maryland.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Pharmaceutical Research and Manufacturers of America (PhRMA) adopted measures to strengthen its Guiding Principles for Direct to Consumer (DTC) Advertisements about Prescription Medicines.

ePT--the Electronic Newsletter of Pharmaceutical Technology
A US Food and Drug Administration guidance issued Tuesday provides new recommendations to applicants who wish to designate proposed products as orally disintegrating tablets (ODTs).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Commission issued proposals last week to improve Europe's pharmaceutical market and patient safety.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration released a draft guidance document that contains questions and answers relating to new labeling requirements for over-the-counter drug (OTC) products that are marketed without an approved application under section 502(x) of the Dietary Supplement and Nonprescription Drug Consumer Protection Act of 2006.

ePT--the Electronic Newsletter of Pharmaceutical Technology
President-elect Barack Obama officially nominated former Senator Tom Daschle as Secretary of Health and Human Services and named him the Director of a new White House Office on Health Care Reform late last week.

Equipment and Processing Report
A surprising amount of the discovery and development life cycle is still based on manual and disconnected process steps.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, NicOx and DSM make manufacture and supply pact for naproxcinod drug substance; NovaRx appointed Norrie Russell president and COO; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration has released Guidance for Industry: Cooperative Manufacturing Arrangements for Licensed Biologics, which finalizes the draft from August 1999.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Environmental Protection Agency is proposing to add hazardous pharmaceutical wastes to the Universal Waste Rule to provide a system for disposing of hazardous pharmaceutical wastes.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Synthetic Chemical Organic Manufacturers Association (SOCMA) has raised concerns over requirements for security vulnerability assessments (SVA) under the US Department of Homeland Security's (DHS) Chemical Facility Anti-Terrorism Act Standards (CFATS).

FDA's Final Rule titled "Amendments to the Current Good Manufacturing Practice Regulations for Finished Pharmaceuticals" takes effect Monday, Dec. 8, 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Reps. John D. Dingell (D-MI), current chairman of the Committee on Energy and Commerce in the US House and Representatives, and Bart Stupak (D-MI), chairman of that committee?s Oversight and Investigations Subcommittee, said that moving the Food and Drug Administration Globalization Act and other measures for drug and food safety will be a key priority for the next Congress.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, AstraZeneca announces changes to its supply chain operations; Christian Velmer appointed head of Wyeth Canada; More...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The European Commission (EC) published a preliminary report stating that competition in the pharmaceutical industry does not work as well as it should.

When accusations fly: 'Tis better to give than to receive.

The past year saw major acquisitions attempted, completed, rejected, and stalled.

Readers provide insight into the best companies to work for as well as the ups and downs of their jobs.
While the world pulls itself out from one of the worst crises in decades, Indian pharmaceutical companies are trying to capitalize on falling company prices by increasing their takeovers.

To expand coverage amidst the economic crisis, Obama will look for ways to cut healthcare costs.

China's quality approach to domestic versus exported products seems to be a lose-lose situation.
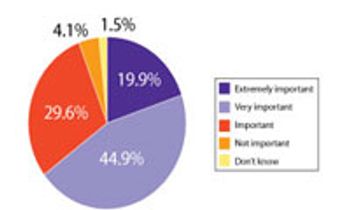
A recent Pharmaceutical Technology survey examined the level, sources, and reasons behind innovation in drug development and manufacturing. This article contains bonus online-exclusive material.
Pharmaceutical Technology is pleased to recognize the winners of its Innovations in Pharma Science Awards.