
An outstanding new book reviews alternative solvents with an eye to sustainable pharmaceutical processes.

An outstanding new book reviews alternative solvents with an eye to sustainable pharmaceutical processes.

A second-generation and green manufacturing process for testosterone provided economic and ecological benefits.
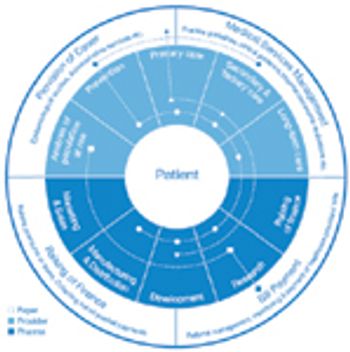
Personalized medicine and integrated healthcare delivery require new business and pricing models. This article contains bonus online-exclusive material.

Representatives of Japan's MHLW report on recent ICH activities and what the ministry expects from Q11.

IPEC's new stability testing guide takes into account the full supply chain's storage conditions.
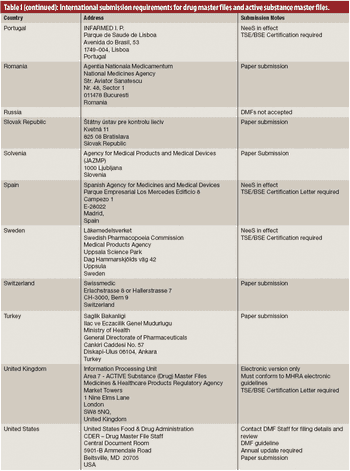
The author describes several issues in creating drug master files and active substance files for active pharmaceutical ingredients and intermediates.

Designated as a "pharmerging market," Brazil is revamping its pricing models.

Pharmaceutical Technology Europe PTE
Jones explains how to save UK pharma.

Pharmaceutical Technology Europe
Merger, union, alliance, partnership, collaboration: five words that ultimately suggest the strengthening of something.

Pharmaceutical Technology Europe PTE
Scientists at Sigma-Aldrich's new Sigma Advanced Genetic Engineering Labs in St Louis (MO, USA) will develop and distribute genetically engineered animal models for use in research.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Eastern Europe's pharmaceutical market is forecast to grow at a CAGR of more than 10% to be worth more than $41 billion by 2014, according to a report from companiesandmarkets.com.

ePT--the Electronic Newsletter of Pharmaceutical Technology
On Aug. 19, 2009, the US Food and Drug Administration opened a new Center for Tobacco Products on the agency's White Oak Campus in Silver Spring, Maryland.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Lonza (Basel) has submitted a non-binding proposal that would see it acquire all of the Restricted Voting Shares of US-based Patheon.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Scientists have modified a tobacco plant to produce a vaccine for norovirus, the viral infection sometimes called the "cruise ship virus."

ePT--the Electronic Newsletter of Pharmaceutical Technology
Researchers at the J. Craig Venter Institute (JCVI), a genomic research organization, reported that they successfully transformed one bacterium into a different strain by transferring the entire bacterial genome of the first strain into a second, related strain of bacteria.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration announced in an Aug. 20, 2009 release that it would like to amend postmarket safety reporting regulations "to require that manufacturers and other facilities subject to current reporting requirements submit their reports in an electronic format."

ePT--the Electronic Newsletter of Pharmaceutical Technology
Company and People Notes: UCB and Novartis form agreement; AAIPharma appoints VP of regulatory affairs; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, USP signed a Memorandum of Understanding with the Permanent Commission of the Pharmacopeia of the United Mexican States, more...

Pharmaceutical Technology Europe PTE
A transatlantic strategic partnership has been struck between California (USA) based enterprise software solution provider ValGenesis and IT integration specialists Systec & Services (Germany).

Pharmaceutical Technology Europe PTE
Researchers from a university in the US are urging the FDA to demand that drug manufacturers state how new medications compare with similar treatments on product labels.

Pharmaceutical Technology Europe PTE
Only 43% of European Chief Financial Officers (CFOs) and Chief Information Officers (CIOs) have tried to calculate the impact of outsourcing on their company, and of those that have tried, only 1 in 5 are confident in their sums, according to a study by Cognizant.

Pharmaceutical Technology Europe PTE
Amcor (Australia) has made an offer of $2025 million for parts of Alcan Packaging (France), including the company's global pharmaceuticals division.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Researchers at the University of Illinois (Champaign) have developed a cancer drug delivery system that reportedly kills target tumor cells, spars healthy cells, and has effects that can be reversed to halt potentially hazardous side effects.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA publishes draft guidances of two ICH Annexes; EMEA sets format for compliance advice; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
Researchers from the Stanford University School of Medicine in California are urging the US Food and Drug Administration to demand that drug manufacturers state how new medications compare with similar treatments on product labels.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration announced a new program regarding the issuance of warning letters to the pharmaceutical industry.

ePT--the Electronic Newsletter of Pharmaceutical Technology
India accounts for approximately 3% of the global outsourcing market, which indicates a significant opportunity for growth, according to a report published by Ernst and Young and the Organization of Pharmaceutical Producers of India.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Only 43% of European chief financial officers (CFOs) and chief information officers (CIOs) have tried to calculate the impact of outsourcing on their company, and of those that have tried, only 1 in 5 are confident about their calculations, according to a study by Cognizant, a global business technology company based in Teaneck, NJ.

ePT--the Electronic Newsletter of Pharmaceutical Technology
An initiative to develop consensus-based industry standards to identify and define green chemicals and process technologies is underway.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, XCELERON and GSK form agreement; Millipore appoints VP of life sciences; more...