
ePT--the Electronic Newsletter of Pharmaceutical Technology
On June 25, US Marshalls seized all drug products and ingredients at three facilities of Caraco Pharmaceutical Laboratories.

ePT--the Electronic Newsletter of Pharmaceutical Technology
On June 25, US Marshalls seized all drug products and ingredients at three facilities of Caraco Pharmaceutical Laboratories.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The UK's Medicines and Healthcare Regulatory Agency (MHRA) has published the outcome of a consultation on measures to strengthen the country's drug supply chain.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Wyeth and Catalyst sign agreement; FDA seeks public opinion about tobacco regulation; Catalent appoints VP of quality and regulatory affairs; more...
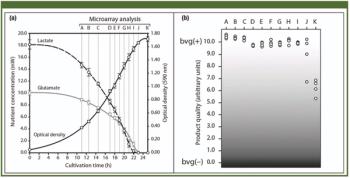
This case study describes the implementation of process analytical technology on the cultivation process step of a whole-cell vaccine against whooping cough disease.

Pan coating has been the preferred method of coating tablets for more than 20 years, but core coating is becoming more popular.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Federal Trade Commission (FTC) last week issued an interim report that examined the effects of authorized generics on competition in the prescription drug market.

ePT--the Electronic Newsletter of Pharmaceutical Technology
This week, the US Pharmacopeial Convention and the Vietnamese Pharmacopoeia Commission signed a memorandum of understanding that will help ensure the safety of Vietnamese medicines.
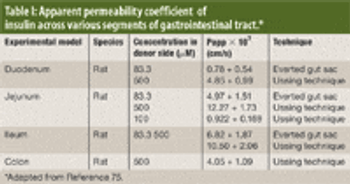
The authors review various oral drug delivery systems that have been explored to increase patient compliance for insulin.

While regulators begin to address nano-based drugs, industry should get its risk data ready.

Some GMP agents seem to find a way to squander time, money, and common sense.

Traditional Chinese Medicine is widely used, but questions persist regarding its regulatory status.
The authors discuss how strategic outsourcing to contract manufacturing organizations that have technical and regulatory expertise can add further value during vaccine development.
Pharmaceutical Technology Europe
Biopharmaceutical companies generally try to avoid paying royalties on novel technologies, including novel expression systems, in part because of the inability to predict revenue flow after a product is commercialized.

Pharmaceutical Technology Europe
Patents are an important tool for protecting innovative products, uses or processes intended for commercialization.
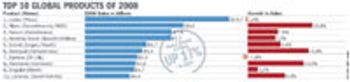
Pharmaceutical Technology Europe
The world's top 50 pharmaceutical companies accounted for prescription drug sales of $558 billion (413 billion euro) in 2008.

Pharmaceutical Technology Europe
Are manufacturers ready to deal with the economic and logistical challenges that accompany tailored therapeutics?

Pharmaceutical Technology Europe
You are invited to Link In and Twitter with PTE!

Pharmaceutical Technology Europe
Rare diseases represent an important area of unmet medical need.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Adimab forms deals with Merck and Roche; Manhattan Pharmaceuticals' CEO and president steps down; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Physician Payments Sunshine Act is still pending before the US Senate Committee on Finance, according to Jill Kozeny, communications director for Senator Chuck Grassley (R-IA).

ePT--the Electronic Newsletter of Pharmaceutical Technology
Generic-drug and specialty pharmaceutical manufacturer Watson Pharmaceuticals (Corona, CA) agreed to acquire the generic-drug company Arrow Group for $1.75 billion in cash and stock.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Former Senator Majority Leaders Howard Baker, Tom Daschle, and Bob Dole released a report this month, Crossing Our Line: Working Together to Reform the US Health System, which proposes four pillars of health reform.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Following the 2008 launch of the European Fine Chemicals Group (EFCG) Voluntary Guidelines (VGs) to promote supply-chain security, the group has now announced the official launch of an assessment template that will allow fine chemicals customers and suppliers to assess and implement this new set of recommendations.

The new commissioner of the US Food and Drug Administration is mapping plans for turning around an agency that has been demoralized, buffeted about in the press and has lost some of the trust of the American people.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, FDA debars clinical investigators; Jubilant Organosys forms deal with Endo Pharmaceuticals; AMRI makes changes to its India management team; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Subcommittee on Health of the US House of Representatives Energy and Commerce Committee held hearings last week to discuss the findings of a report by the Federal Trade Commission (FTC) that examined the competitive effects for follow-on-biologics (FOBs).

ePT--the Electronic Newsletter of Pharmaceutical Technology
Last week, the steering committee and expert working groups of the International Conference on Harmonization met in Yokohama, Japan, to discuss pending guidelines and other relevant issues.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Following the World Health Organization's declaration last week of an influenza pandemic, vaccine makers in Europe and China as well as world health agencies are stepping up efforts toward rapid development and approval of an effective vaccine.

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment & Processing Report, reviews the Editor's Picks for the May 2009 edition from Bullard and Teledyne Tekmar.