
Although industry is tightening its belt, contract manufacturers across Europe are actually making out quite well by taking on additional projects and new roles.

Although industry is tightening its belt, contract manufacturers across Europe are actually making out quite well by taking on additional projects and new roles.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Senator Charles Grassley (R-IA) sent a letter to Frank Torti, acting commissioner of the US Food and Drug Administration, to express concern about a memo that Torti sent to agency staff.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, SOCMA changes name; two FDA approvals; Biogen Idec names chief operating officer; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
On March 26, the Michigan House of Representatives passed House Bill 4316, effectively repealing part of a 1996 law that provides drug companies immunity from liability lawsuits involving products that have been approved by the US Food and Drug Administration.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The United Kingdom's Medicines and Healthcare products Regulatory Agency (MHRA) seized nearly half a million pounds worth of counterfeit medicines on Mar. 26, 2009 in Middlesbrough, England, according to a MHRA press release.

In a recent book, UK regulators explain how to establish a pharmacovigilance system.

Standards data is helpful, but FDA needs to apply its information across the board. This article contains bonus online-exclusive material.

Obama's cost-containment and science-innovation initiatives need to overlap.

President Obama's economic recovery plain includes goals such as reducing the number of uninsured citizens and improving the quality of healthcare.

The financial and economic downturn is likey to have long-term implications for outsourcing.

The role of automation suppliers is transforming to meet the pharma industry's demand for change.

GMP agents report on old products, aseptic violations, and unexpected emotions.

Pharmaceutical Technology Europe
The formation of a partnership, whether personal or professional, is risky business.

ePT--the Electronic Newsletter of Pharmaceutical Technology
President Obama's plan for increasing drug comparative effectiveness research is moving forward. The 15 members of the Federal Coordinating Council for Comparative Effectiveness Research, created to manage the $1.1 billion allocation designated for the research in Obama's American Recovery and Reinvestment Act of 2009, were named last week, according to a Mar. 19 press release from the US Department of Health and Human Services

ePT--the Electronic Newsletter of Pharmaceutical Technology
On March 19, 2009, IMS, the leading provider of market research for the pharmaceutical and healthcare industries, reported that annual sales of prescriptions in the US grew 1.3% from 287.6 billion in 2007 to 291.5 billion in 2008.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Hospira to reduce workforce; WuXi AppTech makes senior appointments; more...

ePT--the Electronic Newsletter of Pharmaceutical Technology
In a case decided on Mar. 20, 2009, the US Court of Appeals for the Federal Circuit invalidated a US Patent and Trademark Office (PTO) Final Rule that governed the number of applications that parties may file to seek continued examinations of patent applications.

ePT--the Electronic Newsletter of Pharmaceutical Technology
President Barack Obama signed the fiscal year 2009 Ombnibus Appropriations Act on Mar. 11, 2009, giving the US Food and Drug Administration $2.6 billion. The funding can be used through the end of the fiscal year, Sept. 30, 2009.

ePT--the Electronic Newsletter of Pharmaceutical Technology
A bipartisan bill that would establish a regulatory pathway for the approval of biosimilars was introduced into the US House of Representatives last week.

Instead of protecting the public from unsafe drugs and contaminated foods, the Food and Drug Administration is a "hazard to public health," stated President Barack Obama in announcing his choices to head the agency and new efforts to improve food safety. Margaret Hamburg will be FDA's new commissioner, and Joshua Sharfstein principal deputy commissioner for drugs and medical products.

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Pharmacopeial Convention (USP) and the National Institute for the Control of Pharmaceutical Biological Products (NICPBP), China's agency for overseeing the quality of large- and small-molecule drugs, signed a memorandum of understanding (MOU) to bolster the quality of medicines in China and in the countries that buy Chinese drug products, including the United States.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Genzyme receives warning letter; Mesa Laboratories appoints John J. Sullivan CEO and a member of the board of directors; more...
Equipment and Processing Report
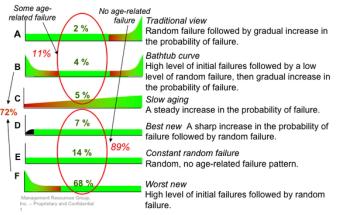
Time-based maintenance programs preserve the equipment but usually not its function, and they do not mitigate equipment failure for the balance of a machine's life cycle.

Equipment and Processing Report
PharmTech's monthly newsletter, Equipment & Processing Report, reviews the Editor's Picks for the February 2009 edition from PortaFab and Sterling.

Equipment and Processing Report
The spotlight on the biopharmaceutical industry is intensifying, as recently evidenced by Pfizer's (New York) ongoing acquisition of Wyeth (Madison, NJ), which was initiated partly to reduce the former's dependence on small-molecule drugs.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Also, Penn Pharma to expand; stem cell research funding ban lifted; Bristol-Myers Squibb made senior appointments; more...

Merck & Co. (Whitehouse Station, NJ) and Schering-Plough (Kenilworth, NJ) completed a definitive merger agreement under which Schering-Plough stockholders will receive $23.61 per share.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Ending a long, closely watched debate over the issue of federal preemption, the US Supreme Court on March 4, to uphold a $6.8 million Vermont Supreme Court decision of Diana Levine against Wyeth Pharmaceuticals (Madison, NJ).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The Synthetic Organic Chemical Manufacturers Association (SOCMA) said last week that Congress is likely to the include inherently safe technology (IST) measures in proposed chemical site-security legislation that is likely to be introduced in late winter or early spring.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Senators Byron Dorgan (D-ND), Olympia Snow (R-ME), John McCain (R-AZ), and Debbit Stabenow (D-MI) introduced a bill March 4 that would allow pharmacists and wholesalers to import prescription drugs from Australia, New Zealand, Japan, Switzerland and the European Union.