
Pressure to approve new user fees opens the door to action on drug shortages, prices, and regulation.

Pressure to approve new user fees opens the door to action on drug shortages, prices, and regulation.

As part of the BRIC bloc with Russia, India, and China, Brazil is one of the world's leading emerging economies and is also considered by IMS Health to be one of seven pharmerging nations, which also include Mexico, Turkey, and South Korea.
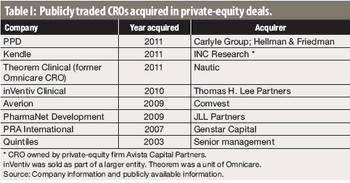
Some recent private-equity buyouts of CROs show both the upside and downside for investors.

The European Union market takes steps toward continuous processing and modular facilities.

A Q&A with Gilles Cottier, president of SAFC, on recent industry trends.
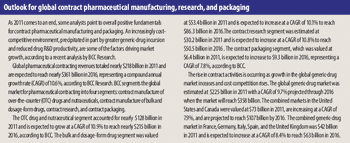
Expansion activity was limited as fine-chemical producers and CMOs of API and intermediates grapple with changing industry fundamentals.

A dearth of late-stage candidates could hurt the pharmaceutical services market in the future.

Cleanliness is crucial, even if zapping and trapping is necessary to reduce product contamination.

Added responsibilities and outside concerns prompt overhaul of agency's structure.

Drug shortages, supply-chain security, generic-drug incursion, and flexible manufacturing models are some of the issues shaping the bio/pharma industry.

Packaging is indeed headed to be a lead sector in the Asian pharmaceutical environment, but certain challenges must first be overcome.

A Q&A with Deborah Tanner, executive vice-president and group president of R&D laboratories at Covance, on recent industry trends.

Readers react to the economic turmoil of the past year and look longingly forward to 2012.

Small drug companies with hopes of achieving $1 billion in sales can pursue various strategies.

Clamor mounts over compromised care and rising costs due to lack of crucial therapies.

Careful mixing during a product's distillation can help avert trouble from a strong concoction.

Researchers develop various catalytic approaches for improving yield, purity, stereoselectivity, and process conditions.
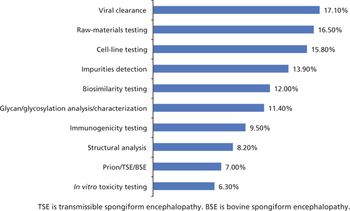
Biosimilar manufacturers need better expression systems and analytical tools to compete.

Russia is aiming to provide an alternative to China and India for drug manufacturing, including APIs.

The authors propose a plan to keep the US pharma industry afloat and in the lead.

Equipment and Processing Report
Recent recalls have contributed to the pharmaceutical industry?s heightened awareness of glass delamination (i.e., the formation of glass flakes in a vial), which could affect drug quality and patient safety. To confront this growing problem effectively, drugmakers must understand its causes.

John Kelly, vice-president of strategy and transitioning sites for Pfizer Global Supply, discusses the company's manufacturing and supply strategy and network.

Getting the most value out of M&As requires proper upfront legwork.

Expanding the organization's mandate will strengthen inspections.

The authors examine risk management relating to the quality issues of clinical-trial materials and discuss areas that would benefit from additional consideration and harmonization.