
The global excipients market shows moderate growth, increased consolidation, and expansion activity in emerging markets and select product areas.

The global excipients market shows moderate growth, increased consolidation, and expansion activity in emerging markets and select product areas.

Pharmaceutical Technology Europe
With the Western world recovering slowly from a recession, and tighter restrictions for medical products being applied by the FDA and other regulatory authorities, more medical device R&D is being located in Asia.

Pharmaceutical Technology Europe
The contract services industry is a crowded market and it's important for companies to differentiate themselves to stand out.

Pharmaceutical Technology Europe
The past year has seen significant consolidation of mid-size CROs, with many of them being bought or put up for sale. In particular, the top six or seven CROs seem to be signing more preferred partner deals, such as Pfizer teaming with Parexel and ICON, and Takeda?s deals with Covance and Quintiles, which is driving mid-size CROs growth through major acquisitions.

Pharmaceutical Technology Europe
Although the European market is approximately 50% smaller than the US in terms of landmass, the population of Europe is approximately 50% larger and this presents a huge market opportunity.

Pharmaceutical Technology Europe
Oriol Prat, Marketing Manager, and Marga Viñes Senior Product Manager, from Grifols give us a quick rundown of how business is changing and what challenges the company faces.

Pharmaceutical Technology Europe
In a quick-fire interview, Dr Stephen Taylor, Vice President & Commercial Director at Fujifilm Diosynth Biotechnologies, looks at some of the challenges facing biologics outsourcing.

A Q&A with John Plachetka, chair, president, and CEO of POZEN, on recent industry trends.

PDUFA renewal legislation sets stage for new policies affecting revenue, resarch, and oversight.

Being aware of a forthcoming inspection or how a product was made can make a huge difference.

Internal and external Web-based communities are changing how pharma companies can innovate.

Growth and change in Brazil and Mexico offer key opportunities for the region's pharmaceutical industry.

The EU debt crisis portends of possible negative repercussions for the dose CMO industry.

The author offers perspectives on ways in which pharmaceutical companies and other stakeholders in the supply chain can confront the threat of counterfeit products, cargo theft, illegal diversion, and economically motivated adulteration.

Biocatalysis, chemocatalysis, and other chiral technologies continue to attract the investment dollars of CMOs and fine-chemical companies.

Pharmaceutical Technology Europe
The increased trend of outsourcing coupled with a relatively strong economy has seen the fine chemicals market grow at a very high level when compared to historical data.

As contract manufacturers and fine-chemical suppliers gather for CPhI/ICSE, effective strategies for technology differentiation are key in an increasingly competitive environment.

A Q&A with Brian Johnson, senior director of supply chain security at Pfizer, moderated by Patricia Van Arnum. Part of a special Ingredients issue.

Last week, Ben Venue Laboratories decided to exit the contract-manufacturing business during the next several years, thus ending more than 70 years of service in this field. To ensure the supply of medically necessary products, the company will work with its customers to develop and execute long-term transition plans.
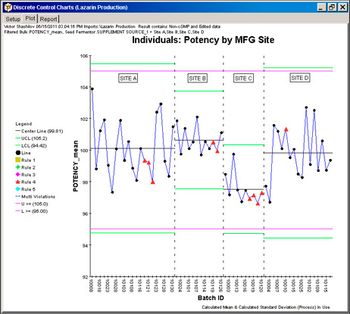
Equipment and Processing Report
Using information technology tools to enhance process understanding helps reduce variability that can affect speed to market.

The Society for Chemical Manufacturers & Affiliates commented on EPA's modifications to the Inventory Update Reporting rule, also broadly know as the Chemical Data Reporting rule. EPA issued the final rule earlier this month.

The International Society for Pharmaceutical Engineering published a guidance document that defines current best practices in pharmaceutical manufacturing applications for handling gases that come into direct contact with the biopharmaceutical and pharmaceutical process steams.

Rising imports and overseas production spur realignment of enforcement.

Contract manufacturing organizations throughout Asia are increasing their capabilities to meet market demand and attract foreign investment and partnerships.
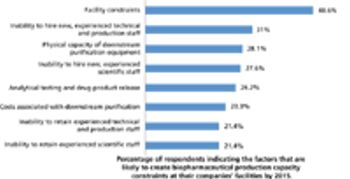
China rises to the top as a destination for international outsourcing.