
Formulation development can involve several complex technical challenges, but an approach incorporating design of experiments can help address these difficulties.

Formulation development can involve several complex technical challenges, but an approach incorporating design of experiments can help address these difficulties.

Using a novel automated microfilling system, the authors demonstrate that roller compaction followed by milling is a viable preprocessing technique for high-dose chemical-in-capsule dosage forms. The process results in higher bulk and tapped densities for drug substances compared with milling alone.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Novartis (Basel, Switzerland) will build a cell culture-derived influenza vaccines manufacturing plant in Holly Springs, North Carolina. Construction is expected to begin in 2007.

ePT--the Electronic Newsletter of Pharmaceutical Technology
In a move to strengthen its position in Western generic drug markets, Ranbaxy Laboratories Ltd. (Gurgaon, Haryana, India) acquired the Mundogen generic drug business of GlaxoSmithKline (GSK, London, England) in Spain, through Ranbaxy's Spanish subsidiary, Laboratorios Ranbaxy S.L.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Although the number of anti-infective vaccines (as distinct from therapeutic vaccines for cancers and other noninfectious diseases) entering clinical study each year since 2000 has been higher on average than it was in the 1990s, this product area may see little additional growth through the rest of this decade, according to a recentanalysis from the Tufts Center for the Study of Drug Development (Boston, MA).

ePT--the Electronic Newsletter of Pharmaceutical Technology
The US Food and Drug Administration (Rockville, MD) announced that Baxter Healthcare Corp. (Deerfield, IL) signed a consent decree relating to the company's "Colleague" volumetric infusion pump and "Syndeo" patient-controlled analgesic syringe pump.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Dutch biotechnology company Crucell NV (Leiden, Netherlands) and its technology partner DSM Biologics BV, a business unit of Royal DSM NV (Heerlen, Netherlands) will open a new research and development center that will specialize on further developing the "PER.C6" human cell line for the expression of recombinant pharmaceutical proteins.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Does the competition from authorized generics really help lower drug prices and boost healthcare savings?
ePT--the Electronic Newsletter of Pharmaceutical Technology
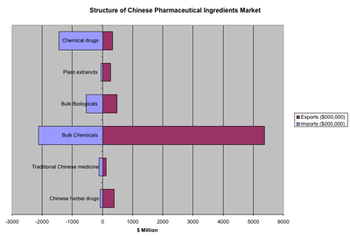
The sixth CPhI China exhibition, presented June 27?29 in Shanghai, offered a showcase for the explosive growth of the Chinese pharmaceutical sector.

University of Buffalo (Buffalo, NY, www.buffalo.edu) researchers have developed a drug delivery system that uses an external magnetic field to guide drug-filled nanocarriers to cultured tumor cells.

ePT--the Electronic Newsletter of Pharmaceutical Technology
MedImmune, Inc. (Gaitherburg, MD) reports that US Food and Drug Administration (Rockville, MD) has approved the company's supplemental biologics license application to use reverse genetics technology to construct new vaccine strains to produce seasonal influenza vaccines.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Biopharmaceutical company Lipoxen PLC (London, UK) has developed a Hepatitis E vaccine using its novel vaccine delivery technology "ImuXen," which the company claims to be easy to manufacture. According to the company, the proprietary liposomal formulation method delivers vaccine materials to the immune system in a manner designed to emulate the response of a natural encounter with the infection agent.

FDA is conducting laboratory research to understand better the ability of preclinical screening tests to identify potential risks and toxicities of nanotechnology-based drugs.

Select large custom manufacturers expand capacity, private equity firms buy companies in transition, and players from India and China build their positions.

Pharmaceutical Technology Europe
The hydrophilic matrix system continues to be the most popular and widely used strategy to achieve extended drug release. Hypromellose (hydroxypropylmethylcellulose [HPMC]) is typically the polymer of choice for the rate-controlling carrier in these systems.

Last week, the Generic Pharmaceutical Association (GPhA Arlington, VA) praised a proposal by the Senate Agricultural Appropriations Subcommittee that, if approved, would provide $10 million in additional funding for the US Food and Drug Administration?s (Rockville, MD) Office of Generic Drugs.

The US Food and Drug Administration (Rockville, MD) approved the first generic versions of "Zocor" (simvastatin), the anticholesterol drug by Merck & Co., Inc. (Whitehouse Station, NJ).

Pfizer, Inc. (New York, NY) reports the United Kingdom's Court of Appeal has upheld the exclusivity of the main patent covering atorvastatin, the active ingredient in "Lipitor."

Drug development opportunities, specification development, and new vaccine technologies were highlighted at the AAPS National Biotechnolgy Conference in Boston this week. More than 1100 attendees from 19 countries participated in the event.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Preformulation represents an early-stage opportunity to facilitate the eventual movement of a drug substance into a commercial product. Strategies to optimize the preformulation process were outlined by Harry Brittain, institute director for the Center for Pharmaceutical Physics (Milford, NJ). He spoke at the PharmTech Annual Event in Somerset, New Jersey this week.

ePT--the Electronic Newsletter of Pharmaceutical Technology
GlaxoSmithKline PLC (GSK, London, UK) will invest more than GBP102 million ($188 million) over the next four years in a vaccine manufacturing plant in Singapore.

ePT--the Electronic Newsletter of Pharmaceutical Technology
RNAi therapeutics company SR Pharma plc (London, UK) has developed a process that allows it proprietary liposomal-based siRNA formulations ?AtuRNAi? drugs to be stored at room temperature and reconstituted in one step.

ePT--the Electronic Newsletter of Pharmaceutical Technology
Recombinomics (Pittsburgh, PA) is again urging the World Health Organization to fully release all H5N1 avian influenza sequences, claiming their release would improve the selection of vaccines by helping scientists to identify the origin of the isolates and predict sequence changes.
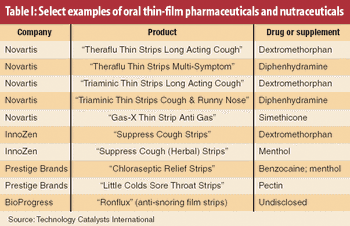
The biggest single recent trend in outsourcing solid-dosage processing has been the movement toward discovery and synthesis of more potent active pharmaceutical ingredients.
Pharmaceutical Technology Europe
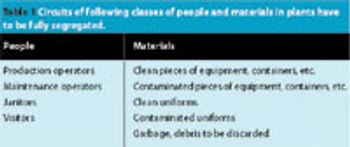
The construction of a new oral solid form (OSF) plant is an important decision and a real challenge. The team in charge of the basic conceptual design has to ensure that the new plant will be up-to-date and efficient not only at start-up, but for the next 15–20 years. This means that the project must be able to adjust to capacity changes, product changes and technology changes. It sometimes seems like an impossible challenge.