
The results of forced degradation studies indicate the need for alternatives to valerophenone as an internal standard calibration for quantifying ibuprofen in bulk drug and tablet assay samples.

The results of forced degradation studies indicate the need for alternatives to valerophenone as an internal standard calibration for quantifying ibuprofen in bulk drug and tablet assay samples.

Pharmaceutical Science & Technology Innovations

Rapid structural elucidation of compounds in complex mixtures is a powerful technique in metabolite, degradation and process control applications. The ability to follow the fragmentation pathway through sequential MSn transitions provides added confirmation and increases the selectivity for monitoring compounds of interest in a complex mixture. Previously, the limiting factors in applying this technique were sensitivity at MS4 and higher transitions, and the cycle time required to acquire multiple MSn spectra across a narrow LC peak. Recent developments in trapping, detection efficiencies and scan rates have reduced these limitations and enabled rapid characterization of multiple compounds from single chromatographic runs.
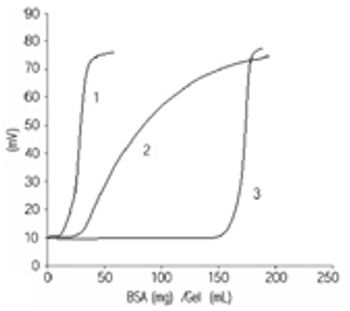
Tosoh has developed a new product - Toyopearl Toyoscreen that allows fast identification of the optimal resin for your sample. You can develop the optimal conditions for the separation in addition to the determination of the dynamic binding capacity. All the resins prepacked in these columns are real process resins concerning particle size. Results on the small columns can be scaled up to production.

Automated actuation of nasal sprays removes operator bias during measurement of particle size, spray-plume geometry, and delivered dose.

The authors outline stability studies designed to evaluate the effect of temperature excursions on product quality that may occur during distribution.

The role of microbial testing to ensure the sterility of aseptically filled sterile products is explained, from the product development phase to in-process monitoring to finished product testing.

Pharmaceutical Science & Technology News

This article introduces the application of high-resolution ultrasonic spectroscopy (HR-US) for the analysis of emulsions and suspensions. The authors outline the principles of the technique and illustrate its application for analysis of the crystallization of lysozyme and the formation of a microemulsion.

The author describes a separation method for two active ingredients in the contraceptive pill with liquid chromatography UV detection.

Current microbiological methods cannot measure microbial contamination at the levels that engineers and regulators seek to establish for aseptic processing cleanrooms. New approaches for assessing data and establishing alert and action levels are advocated, and an example of one analytical tool is considered.

Dry powder inhalers are a well-accepted dosage form for pulmonary drug delivery and a wide variety are either currently available or in development. This article examines a premetered, capsule-based multidose inhaler for which different qualities of a-lactose monohydrate were screened.

The bioavailability of some insoluble drugs is enhanced when they are dissolved in the solubilizing agent macrogol 400, although conventional hard capsules cannot tolerate the agent. This article investigates a PVA copolymer, which has been developed by the authors, examining its properties and its suitability as a material in capsule formulations.

Cationic liposomes are widely used in gene therapy as a safe alternative to highly immunogenic viral vectors. Attachment of a tissue-specific ligand to the surface of the liposomes can increase specificity and reduce undesired transfection. Targeted liposomes can be categorized as either immunoliposomes or ligand-targeted liposomes. The author provides a brief review of tumour-specific and liver-targeted cationic liposomes and strategies for the development of liposome?ligand complexes.

This article reflects on the challenges that predicting powder flowability currently pose to the pharmaceutical manufacturing industry and considers some of the benefits that can accrue when companies overcome these issues.

The use of solid dispersion technology to increase the bioavailability of poorly water-soluble drugs has always been limited by processing and scale-up difficulties. A new approach may help to overcome some of the problems.

This case study describes how a major pharmaceutical manufacturer was equipped with four filling lines, for metered dose inhalers, supplied with a nitrogen cooling system to prevent spontaneous vaporization of the propellant gas. By doing so, a cost-effective and environmentally friendly solution was provided to a hazardous situation, which also complied with regulatory directives.
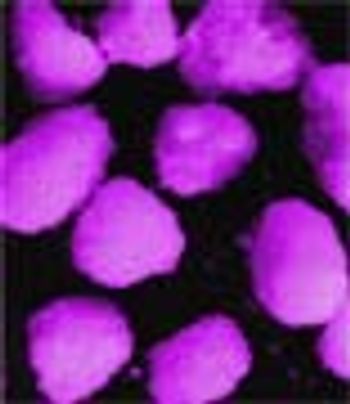
Previous articles have presented a general review of the different types of spheres that can be obtained with a rotary fluidized bed process.1,2 This two-part study focusses on lipid spheres that can be prepared using hydrogenated castor oil, as well as examining the feasibility of the process and the main characteristics of the spheres obtained.

This article describes a method for assessing the similarity of dissolution profiles using Hotelling's T2 statistic. The method applies a covariance structure that accounts for the heterogeneity of variance and correlation across time points. Comparing the method with the f2 criterion recommended in FDA's guidance on dissolution testing, the performance of the two methods was assessed on real examples, and simulation studies were also done to compare the method's performance with that of the f2 criterion.

Oral dosage forms are the most popular way of taking medication, despite having some disadvantages compared with other methods. One such disadvantage is the risk of slow absorption of the active pharmaceutical ingredient (API), which can be overcome by administering the drug in liquid form and, therefore, possibly allowing the use of a lower dosage.

Interest in more advanced drug delivery systems has increased, with an acceleration in the discovery and development of novel therapeutic macromolecules for targeted applications. Computational fluid dynamics is a design tool that allows producers of these and other products to evaluate different models rapidly and cost-effectively.

Analytical methods development and validation play important roles in the discovery, development, and manufacture of pharmaceuticals. The official test methods that result from these processes are used by quality control laboratories to ensure the identity, purity, potency, and performance of drug products.

Non-toxic and biodegradeable, biologically active liposomes encapsulate therapeutics to provide an attractive drug delivery system for the future.


Brussels report