
Compounding Pharmacies' Lawsuit Against FDA Will Continue

Compounding Pharmacies' Lawsuit Against FDA Will Continue

FDA Drug Safety Oversight Board

It Helps to be Flexible: Supple WIN Compounds Could Cure the Common Cold

Cytogen and Dowpharm to Develop Targeted Anticancer Product


Bedford Recall

GSK and FDA Agree on Consent Decree
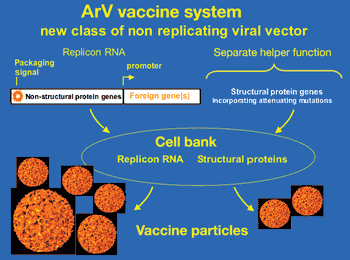
Vaccine developers are using novel drug delivery methods that offer advantages over traditional techniques such as improved immunogenicity, better stability, specific control over antigen release, and a wider pool of targeted diseases.

Vaccine developers are wrking on new drug delivery systems that offer improved immune responses, better stability, and a wider pool of targeted diseases.

The authors review the solution and powder properties of polyethylene oxide and its various applications in hydrogels and hydrophilic matrix systems.

The unit-dose bar coding rule requires integrating many aspects of packaging design and control.

FDA is finalizing guidances and enforcing compliance with manufacturing standards as part of its efforts to ensure drug safety.

A newly developed software program transforms written SOPs for all required analytical method validation experiments into transferable automated templates, integrating individual activities and technologies under one platform.

Major pharmaceutical companies are focusing more than ever on the prices CROs are charging for their services.

New technologies and improvements to existing ones can reduce contamination risk in aseptic processing.

A ready-to-fill closed vial can improve aseptic filling quality and reduce process complexity.

The complete elimination of human-derived contamination is possible only with the elimination of human intervention.

Pfizer Recalls Lot of Neurontin

Crucell and NIH Sign Ebola Vaccine Manufacturing Contract

The Empire Strikes Back: Innovators Releasing Generic Drugs

Warning Letter: Compounding at Palace Pharmacy

The authors propose a strategy for classifying and validating inprocess testing methods.

The author proposes a new analytical graphic, the sector chart, which presents data that cannot be adequately presented with current graphs.This chart combines features of zone charts with the basic principle of precontrol charts. It addresses engineering control and is superior for representing data such as beneficial and adverse trends.

The validation of alternative microbiological testing is an opportunity for a manufacturer to decrease the amount of time required for laboratory results.To properly validate these alternatives, a practitioner must first identify what is being studied. The regulatory effect on established product and process specifications and levels must be completely evaluated, as changing the method of analysis may well change the pparent number in the sample.

RFID and battery-powered containers are among the developments receiving attention in the pharmaceutical cold chain.