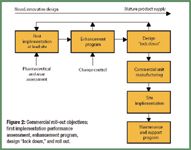
Pharmaceutical Technology Europe
The increasing demand for speed-to-market requires that modern pharmaceutical laboratories work faster, more accurately and efficiently than before. Unavoidably, this leads to a number of bottlenecks that threaten the overall laboratory operation and efficiency.