
Pharmaceutical Technology's In the Lab eNewsletter
The acquisition will increase Metrohm’s spectroscopy offerings and capabilities.

Pharmaceutical Technology's In the Lab eNewsletter
The acquisition will increase Metrohm’s spectroscopy offerings and capabilities.
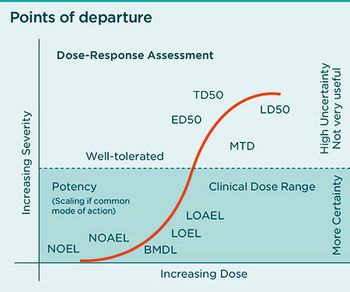
Determining how much containment is needed for API handling requires evaluation of multiple factors.

Pharmaceutical Technology's In the Lab eNewsletter
The National Science Foundation grant will be used to commercialize a synthetic biology platform for cancer drug development.

Pharmaceutical Technology's In the Lab eNewsletter
Waters’ acquisition of Prosolia’s DESI technology boosts its mass spectrometry imaging portfolio.

The agency has issued a warning letter for cGMP violations at a drug manufacturing facility in Ahmedabad, India, that Baxter gained through its acquisition of Claris Injectables.

FDA and ICH seek comment on new exposure levels for cadmium in drug products.
The delivery device and drug form should be considered when choosing a test method for identifying and measuring particulates in inhaled drug products.

Pharmaceutical Technology's In the Lab eNewsletter
Sciex’s latest additions to its biopharma portfolio helps to optimize workflow for better analytics in the lab.

Pharmaceutical Technology's In the Lab eNewsletter
Thermo Fisher Scientific’s mass spectrometer combines mass-analyzer technology with an intelligent data acquisition strategy.

Pharmaceutical Technology's In the Lab eNewsletter
The company’s new LCMS-9030 system is designed for high resolution and accurate mass detection.

Pharmaceutical Technology's In the Lab eNewsletter
The company unveiled its latest innovations to its mass spectrometry portfolio at ASMS in San Diego.

The Analysette 28 ImageSizer from Fritsch provides analysis of particle shape and size for dry measurement of powders and bulk solids and wet measurement of suspensions and emulsions.

Traditional barriers between upstream and downstream bioprocessing are slowly starting to break down, as biopharma embraces more advanced analytics and process control.

Macromolecular drugs are typically injected, but oral dosage forms are being developed to improve the treatment of gastrointestinal conditions such as inflammatory bowel disease.

Pharmaceutical Technology's In the Lab eNewsletter
The company’s next-generation ultraperformance liquid chromatography platform is designed to meet the evolving laboratory requirements for chromatographic performance.

Flow imaging microscopy can be used to identify particulates and their sources.

Open communication channels between all the parties involved is key when outsourcing analytical methods, says Susan Schniepp, distinguished fellow at Regulatory Compliance Associates.

This paper analyzes the effectiveness of histogram binning for representing particle-sizing measurements with accuracy and precision.

Pharmaceutical Technology's In the Lab eNewsletter
National Institutes of Health (NIH) researchers combine two microscope technologies to create sharper, faster images.

Pharmaceutical Technology's In the Lab eNewsletter
The new kit is expected to detect a broad spectrum of pyrogens while offering high reproducibility and sensitivity as well as eliminating the need for live animal testing.

Pharmaceutical Technology's In the Lab eNewsletter
The Amgen Foundation and Harvard University will develop LabXchange, a free online science education platform that will provide digital instruction and virtual lab experiences.

Pharmaceutical Technology's In the Lab eNewsletter
Agilent Technologies expects the acquisition to improve its current software portfolio.

Pharmaceutical Technology's In the Lab eNewsletter
The company’s new UV-1900 UV-VIS spectrophotometer incorporates features that improve usability, regulatory compliance, and performance.

The sterility test isolator (STISO) for testing aseptically filled drug products and for testing toxic aseptic drugs (STISOtox) from Metall+Plastic, a subsidiary of Optima, are modular and ergonomically designed.

The combination of translatable in-vitro and in-vivo assays, coupled with expertise in immunology and oncology helps facilitate the progression of novel oncology therapeutics from hit identification to investigational new drug submission.